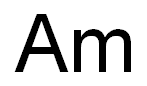AMERICIUM, Chemical element, symbol Am, at no. 95, at. wt. 243 (mass number of the most stable isotope), radioactive metal of the actinide series, also one of the transuranium elements. All isotopes of americium are radioactive; all must be produced synthetically. The element was discovered by G.T. Seaborg and associates at the Metallurgical Laboratory of the University of Chicago in 1945. At that time, the element was obtained by bombarding uranium-238 with helium ions to produce 241Am, which has a half-life of 475 years. Subsequently, 241Am has been produced by bombardment of plutonium-241 with neutrons in a nuclear reactor. 243Am is the most stable isotope, an alpha emitter with a half-life of 7950 years. Other known isotopes are 237Am,238 Am,240 Am,241Am,242Am,244 Am,245 Am, and 246Am.
This element exists in acidic aqueous solution in the (III), (IV), (V), and (VI) oxidation states with the ionic species probably corresponding to Am3+, Am4+, AmO2 + and AmO2 2+. The colors of the ions are: Am3+, pink; Am4+, rose; AmO2 +, yellow; and AmO2 2+, rum-colored.
silvery metal with two forms; α-Am: hexagonal, a=0.3468 nm, c=1.1241 nm; β-Am: cub, a=0.4894 nm; t1/2 241Am=433 years, t1/2242Am=152 h, t1/2 243Am=7400 years; Am+++ stable in aqueous solution; enthalpy of vaporization 230 kJ/mol; enthalpy of fusion 14.4 kJ/mol; enthalpy of sublimation 276 kJ/mol; ionic radius of Am+++ is 0.0982nm; can be prepared from 241Pu; used to diagnose thyroid disorders [KIR78] [CIC73]

All the isotopes of americium belonging to the transuranic subseries of the actinide seriesare radioactive and are artificially produced. Americium has similar chemical and physicalcharacteristics and is homologous to europium, located just above it in the rare-earth (lanthanide)series on the periodic table. It is a bright-white malleable heavy metal that is somewhatsimilar to lead. Americium’s melting point is 1,176°C, its boiling point is 2,607°C, and itsdensity is 13.68g/cm3.
There are 24 isotopes of americium. All are radioactive with half-lives rangingfrom 72 microseconds to over 7,000 years. Five of americium’s isotopes are fissionablewith spontaneous alpha decay.
Named after the continent America because Europium was named
after the European continent.
Americium does not exist in nature. All of its isotopes are man-made and radioactive.Americium-241 is produced by bombarding plutonium-239 with high-energy neutrons,resulting in the isotope plutonium-240 that again is bombarded with neutrons and resultsin the formation of plutonium-241, which in turn finally decays into americium-241 by theprocess of beta decay. Both americium-241 and americium-243 are produced within nuclearreactors. The reaction is as follows: 239Pu + (neutron and λ gamma rays) → 240Pu + (neutronand λ gamma rays) → 241Pu→ 241Am + beta minus (β-); followed by 241Am→ 93Np-237 +4He2 (helium nuclei).
Although americium’s main valence (oxidation state) is +3, it is tetravalent. It can formcompounds with its ions of +4, +5, and +6, particularly when oxidized. Its most stable isotopeis americium-243, with a half-life of 7,379 years, which, over time through alpha decay,transmutates into neptunium-239.
Americium is a transuranic radioactive chemical element located in the periodic table below the lanthanide element europium, and thus by analogy named after another continent, America. However, it was discvered in Earth having been produced for millions of years in natural nuclear reactors in Oklo, Gabon. These ceased to function a billion years ago, and as the longest lived isotope is americium-247, with a half-life of 7370 years, none has survived to the present day. Americium was discovered in 1944 by the group of Glenn T. Seaborg. They produced americium by bombarding plutonium 239 with high energy neutrons. This formed 240Pu, which was itself bombarded with neutrons and changed into 241Pu, which then decayed into 241Am through beta decay. This work was carried out at the University of Chicago's Metallurgical Laboratory, now known as Argonne National Laboratory. The discovery of americium was announced only in 1945 because during the Manhattan project all discoveries were classified (Kostecka, 2008).
Gamma radiography, radiochemical research, diagnostic aid, electronic devices.
Americium-141, with a half-life of 432 years, which produces both alpha (helium nuclei)and gamma rays (somewhat like X-rays), has found some commercial uses. One use is as thedetector used in household smoke alarms The americium-141 isotope emits alpha particlesthat ionize air by removing electrons from the molecules of air and improving its electricalconductivity. Smoke reduces this conductivity, thus triggering the alarm. Another use is americium-241’s high-energy gamma rays in radiography, in which portable X-ray instruments caneasily be transported for emergency use. As a gamma ray source, americium-241 is used as adiagnostic aid to check the quality of welds in metals.
A highly
toxic radioactive silvery element of the
actinoid series of metals. A transuranic element,
it is not found naturally on Earth but
is synthesized from plutonium. The element
can be obtained by reducing the trifluoride
with barium metal. It reacts with
oxygen, steam, and acids. 241Am has been
used in gamma-ray radiography.
Symbol: Am; m.p. 1172°C; b.p.
2607°C; r.d. 13.67 (20°C); p.n. 95; most
stable isotope 243Am (half-life 7.37 × 103
years).
americium: Symbol Am. A radioactivemetallic transuranic elementbelonging to the actinoids; a.n. 95;mass number of most stable isotope243 (half-life 7.95 × 103 years); r.d.13.67 (20°C); m.p. 994 ± 4°C; b.p.2607°C. Ten isotopes are known. Theelement was discovered by G. T.Seaborg and associates in 1945, whoobtained it by bombarding uranium–238 with alpha particles.
Am. A synthetic radioactive element of atomic number 95, a member of the actinide series; aw 241; 14 isotopes of widely varying half-life; valence 3, but divalent, tetravalent, and higher valencies exist. α and γ emitter, forms compounds with oxygen, halides, lithium, etc. Metallic americium is silver-white crystalline, d 13.6, mp approximately 100C. Half-life of 241 Am is 458 years.
All the isotopes and compounds of americium are deadly sources of radiation and causeradiation poisoning and death. Precautions must be taken when working with it. The smallamount of americium-241 found in smoke detectors in household smoke alarms is harmlessunless the isotope is removed and swallowed.