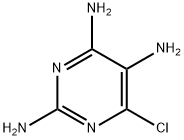
General procedure for the synthesis of 2,4,5-triamino-6-chloropyrimidine from 2,5-diamino-4,6-dichloropyrimidine: 2,5-diamino-4,6-dichloropyrimidine (1 equiv.) was suspended in concentrated ammonia (18 mL/mmol), placed in a sealed reaction tube, and heated to 135 °C for 24 hours. Upon completion of the reaction, the reaction mixture was concentrated under vacuum and subsequently purified by silica gel column chromatography with the eluent being a solvent mixture of dichloromethane/methanol/concentrated ammonia to give the final target compound 2,4,5-triamino-6-chloropyrimidine (93% yield). The structure of the product was confirmed by 13C NMR (101 MHz, J6-DMSO) with the chemical shifts of δ 156.0, 155.5, 113.3, 39.5, respectively; the analytical results of high-resolution mass spectrometry (HRMS-ESI) were in accordance with the theoretical values, with the calculated value of C4H7ClN5 [M + H]+ 160.03845 and the measured value of 160.0385.