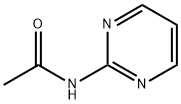
The general procedure for the synthesis of 2-acetylaminopyrimidine from acetamide and 2-chloropyrimidine was as follows: to a screw-capped vial were added acetamide (121 mg, 1.00 mmol), Cs2CO3 (456 mg, 1.40 mmol), Xantphos (87 mg, 0.15 mmol), Pd2(dba)3 (46 mg, 0.050 mmol ), 2-chloropyrimidine (137 mg, 1.20 mmol), and 1,4-dioxane (2 mL). The reaction mixture was deoxygenated by nitrogen bubbling for 3 min, followed by stirring the reaction for 16 h at 100 °C. After completion of the reaction, the mixture was cooled to room temperature. The reaction mixture was diluted with dichloromethane and filtered through a diatomaceous earth pad to remove insoluble impurities. The filtrate was concentrated under reduced pressure to remove the solvent to give the crude product. The crude product was purified by rapid chromatography on silica gel (eluent: 40-100% ethyl acetate/hexane gradient) to give 2-acetylaminopyrimidine (188 mg, 0.85 mmol, 94% yield) as an amorphous solid.