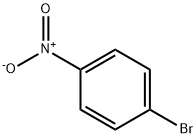
General procedure for the synthesis of 2-nitro-5-bromoaniline from 1-bromo-4-nitrobenzene:
Step 1: Synthesis of 5-bromo-2-nitroaniline (142)
[0745] To a solution of ethylene glycol dimethyl ether (170 mL) containing potassium tert-butoxide (21.92 g, 195 mmol) and copper(I) chloride (2.36 g, 23.8 mmol) was added dropwise a solution of 1-bromo-4-nitrobenzene (141, 8.69 g, 43.0 mmol) in DMF (45 mL) at 0 °C and protected by nitrogen, the time of dropwise addition was 50 min. After the dropwise addition, the cooling bath was removed and the reaction mixture was continued to be stirred at room temperature for 4 hours. After completion of the reaction, the reaction solution was diluted with dichloromethane, washed sequentially with aqueous ammonium chloride, dried over anhydrous magnesium sulfate, filtered and concentrated to give the target compound 142 (7.85 g, 84% yield).
[0746] 1H NMR (CDCl3) δ (ppm): 7.98 (d, J = 9.2 Hz, 1H), 7.02 (d, J = 2.0 Hz, 1H), 6.82 (dd, J = 9.2, 2.0 Hz, 1H), 6.12 (bs, 2H).