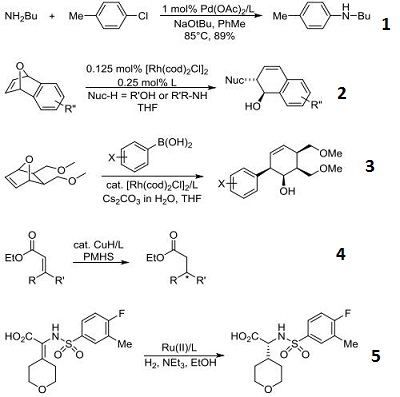
(1) Synthesis of (R)-1-ferrocenyl ethyl di-tert-butylphosphine
Under argon protection, vinyl ferrocene (1 mol, 212 g), (R)-3,3'-bis(3,5-dimethylphenyl)-1,1'-binaphthol phosphonate (0.01 mol, 5.6g) and 1L of toluene, then add di-tert-butylphosphine (1 mol, 147g), heating to 60 reaction for 12 hours, and then cool down and cool down, to the system dropwise addition of water, and then partitioning, the organic layer with anhydrous magnesium sulfate drying, filtration, distillation under reduced pressure to remove the solvent to get a yellow solid, dichloromethane and methanol recrystallization to get (R)-1 - ferrocenylethyl di-tert-butylphosphine 347g, yield 97%, e.e. value 99.5%.
(2) Synthesis of (R)-(-)-1-[(S)-2-(diphenylphosphino)ferrocenyl]ethyldi-tert-butylphosphine
Under the protection of argon, (R)-1-ferrocenyl ethyldi-tert-butylphosphine (0.9 mol, 321 g) and 1 L of ethyl ether were added to a dry reactor, and then a 1 M diethylzinc hexane solution (0.9 mol, 0.9 L ), after the reaction at 0C for 3 hours, diphenylphosphonium chloride (0.9 mol, 199 g) was added dropwise to the system, and after the dropwise addition was completed, it was heated and refluxed for 2 hours, and quenched by adding water to the system, and then partitioned, and the organic layer was dried with anhydrous magnesium sulfate, filtered, and the yellow solid was obtained after the solvent was removed by distillation under reduced pressure, and recrystallized from methylene chloride and methanol to obtain (R)-(-)-1-[(S)-2- diphenylphosphine Ferrocene ethyl-di-tert-butylphosphorus 468g, yield 96%, e.e. value 99.3%.
![(R)-1-[(1S)-2-(DIPHENYLPHOSPHINO)FERROCENYL]ETHYLDI-TERT-BUTYLPHOSPHINE Structure](http://CAS/GIF/155830-69-6.gif)

![(R)-1-[(1S)-2-(DIPHENYLPHOSPHINO)FERROCENYL]ETHYLDI-TERT-BUTYLPHOSPHINE pictures](/ProductImageEN/2021-11/Small/f5db0c88-3f44-48f0-9f1f-4bc724a5cdec.jpg)
![(R)-(-)-[(S)-2-(Diphenylphosphino(ferrocenyl]ethyldi-tert-butylphosphine pictures](/ProductImageEN1/2025-06/Small/1145e017-b834-4040-a4cb-3cc031091a64.png)
![(R)-1-[(SP)-2-(Diphenylphosphino)ferrocenyl]ethyldi-tert-butylphosphine pictures](/ProductImageEN/2021-06/Small/af828add-e549-4bd9-83a1-5b42da7d6026.jpg)