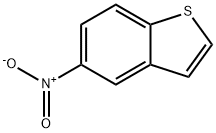
General procedure for the synthesis of 5-aminobenzo[b]thiophene from 5-nitrobenzo[b]thiophene: A mixture of 5-nitrobenzo[b]thiophene (3.09 g, 17.0 mmol) with 10% palladium carbon catalyst (Aldrich, Catalog No. 20,569-9) (150 mg) in ethanol (90 mL) was placed in a Parr reaction flask. The hydrogenation reaction was carried out under hydrogen atmosphere at 3 bar with continuous shaking for 16 hours. Upon completion of the reaction, the catalyst was removed by filtration through a diatomaceous earth pad and the filter cake was washed with ethanol (2 x 30 mL). The combined filtrates were concentrated under reduced pressure to afford benzo[b]thiophen-5-amine (2.57 g, 100% yield) as a dark purple amorphous solid. Its 1H NMR (CDCl3, 400 MHz) data were as follows: δ 3.70 (broad single peak, 2H), 6.78 (double peak, J = 8.61, 1.96 Hz, 1H), 7.10 (double peak, J = 2.35 Hz, 1H), 7.14 (double peak, J = 5.09 Hz, 1H), 7.38 (double peak, J = 5.48 Hz, 1H), and 7.63 (double peak, J = 8.61 Hz, 1H).