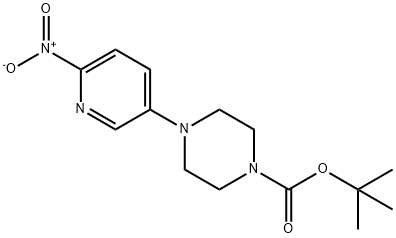
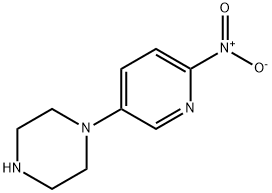
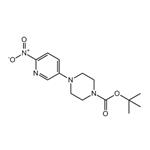
1-(6-nitropyridin-3-yl)piperazine (15.6 g, 0.075 mol) was suspended in 120 mL of tetrahydrofuran (THF) under nitrogen protection. Triethylamine (10.5 mL, 0.075 mol) and 4-dimethylaminopyridine (0.46 g, 5 mol%) were added sequentially to the suspension. Di-tert-butyl dicarbonate (16.6 g, 0.075 mol) was dissolved in 50 mL of THF and transferred to a dropping funnel. The solution was slowly added dropwise to the stirred suspension, and the rate of dropwise acceleration was controlled to maintain the reaction temperature below 27 °C. After dropwise addition, the reaction mixture was cooled to room temperature and subsequently heated to reflux. After refluxing the reaction for 1 hour, a small amount of insoluble material was removed by filtration. The solvent was removed by concentration under reduced pressure and the yellow residue was partitioned between ethyl acetate (EtOAc) and water. The aqueous phase was extracted twice with EtOAc. The organic layers were combined, washed sequentially with a small amount of water and saturated sodium chloride solution, dried and concentrated under reduced pressure. The resulting solid was recrystallized by 2-propanol (treated with activated charcoal) to give the target product tert-butyl 4-(6-nitro-3-pyridinyl)-1-piperazinecarboxylate, ca. 19 g (yield ca. 80%). Nuclear magnetic resonance hydrogen spectrum (1H NMR, CDCl3) δ: 1.49 (s, 9H), 3.46 (m, 4H), 3.65 (m, 4H), 7.20 (m, 1H), 8.17 (m, 2H). Mass spectrum (m/z): 309.