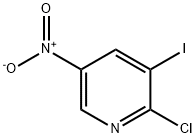
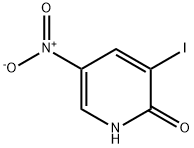
General procedure for the synthesis of 2-chloro-3-iodo-5-nitropyridine from 3-iodo-2-hydroxy-5-nitropyridine:
1. a mixture of 3-iodo-5-nitropyridin-2-ol (37.60 mmol, 10 g), phosphorus trichloride (POCl3, 86.47 mmol, 7.94 mL), and phosphorus pentachloride (PCl5, 48.87 mmol, 10.2 g) was heated under argon protection for 45 min at 140 °C.
2. After completion of the reaction, the mixture was cooled to room temperature, slowly poured into ice water and extracted with dichloromethane (DCM).
3. The organic phase was washed sequentially with water, aqueous sodium bicarbonate (NaHCO3) and brine.
4. The solvent was removed by evaporation and the crude product was purified by column chromatography on silica gel (SiO2), eluting with a hexane/dichloromethane solvent mixture to afford the target product, 2-chloro-3-iodo-5-nitropyridine (7.32 g, 69% yield). 5. The structure of the product was determined by 1H NPD analysis.
5. The structure of the product was confirmed by 1H NMR (300 MHz, CDCl3): δ 8.90 (s, 1H), 9.19 (s, 1H).
Note: Repeat steps have been combined to ensure conciseness and accuracy of experimental steps.