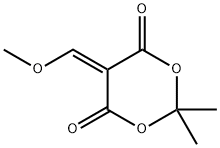
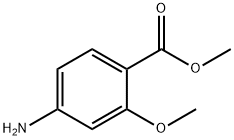
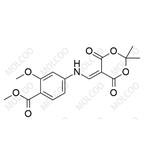
The general procedure for the synthesis of methyl 4-((2,2-dimethyl-4,6-dioxo-1,3-dioxan-5-yl)methylamino)-2-methoxybenzoate from 5-(methoxymethylenyl)-2,2-dimethyl-1,3-dioxane-4,6-dione and 2-methoxy-4-aminobenzoic acid methyl ester was as follows: first, 2-methoxy-4-aminobenzoic acid methyl ester (14.15 g, 78 mmol) was suspended in isopropanol and heated at 50 °C. Subsequently, 5-(methoxymethylenyl)-2,2-dimethyl-1,3-dioxohexane-4,6-dione (14.8 g, 80 mmol) was added in batches over 10 min. The reaction mixture was heated to reflux for 30 minutes. After completion of the reaction, the mixture was cooled to room temperature and the precipitate formed was collected by filtration. The precipitate was washed with isopropanol and dried under vacuum to afford the target product 5-((3-methoxy-4-methoxycarbonylanilino)methylene)-2,2-dimethyl-1,3-dioxane-4,6-dione (25.2 g, 96% yield).
![4-[(2,2-Dimethyl-4,6-dioxo-[1,3]dioxan-5-ylidenemethyl)-amino]-2-methoxy-benzoic acid methyl ester Structure](/CAS/20180703/GIF/205448-64-2.gif)
![4-[(2,2-Dimethyl-4,6-dioxo-[1,3]-dioxan-5-ylidenemethyl)-amino]-2-methoxy-benzoic acid methyl ester pictures](/ProductImageEN/2023-12/Small/7be7b66f-93eb-479e-a665-4f0bf979a9da.jpg)