Under nitrogen protection, 25 mL of triethylamine, 1.415 g (5.0 mmol) of 4-bromoiodobenzene, 420 mg (5.0 mmol) of 2-methyl-3-butyn-2-ol, and 70 mg (0.1 mmol) of bis(triphenylphosphine)palladium dichloride (Pd(PPh3)2Cl2), triphenylphosphine ( PPh3) 105 mg (0.4 mmol), copper iodide (CuI) 38 mg (0.2 mmol), 35 reaction until thin layer chromatography monitoring raw materials on the disappearance of bromoiodobenzene spots as the end point. Reaction solution evaporated solvent under reduced pressure, CH2Cl2 dissolved, washed sequentially with 5% dilute hydrochloric acid, saturated NaCl solution, anhydrous MgSO4 drying, filtration. After the filtrate was evaporated the solvent under reduced pressure, the crude product was purified by column chromatography (eluent: petroleum ether:ethyl acetate=10:1) to obtain 4-(4-bromophenyl)-2-methyl-3-butynyl-2-ol as white solid 1.052 g (4.4 mmol) in 90% yield.
.
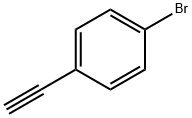
Under the protection of nitrogen, 30 mL of toluene, 4-(4-bromophenyl)-2-methyl-3-butynyl-2-alcohol 478 mg (2.0 mmol), NaOH 480 mg (12 mmol) and diethylene glycol monoethyl ether 8.6 mL were added to the Schlenk reaction flask, and the reaction was carried out at 75 until the disappearance of spots of raw materials was monitored by thin-layer chromatography as the end point. Cooled to room temperature, washed sequentially with 5% dilute hydrochloric acid, saturated NaCl solution, anhydrous MgSO4 drying, filtration. After the filtrate was evaporated to dry the solvent under reduced pressure, the crude product was purified by column chromatography (eluent petroleum ether) to give a white solid 319 mg (1.76 mmol). NMR spectra proved that the product was (4-bromophenyl)acetylene in 80% yield.