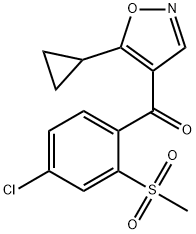
Isoxachlortole is produced commercially through a multi-step synthesis that builds its isoxazole herbicide structure. The process begins with the preparation of the key diketone intermediate, 1-(4-chloro-2-methylsulfonylphenyl)-3-cyclopropyl-2-ethoxymethylenepropan-1,3-dione. This intermediate is then reacted with hydroxylamine hydrochloride in ethanol, under mild conditions with sodium acetate as a base, to cyclise and form the isoxazole ring. After overnight stirring, the mixture is evaporated, extracted with ethyl acetate, washed, dried, and filtered. The crude product is purified by recrystallisation, yielding the final compound.