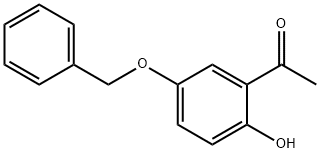
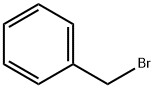
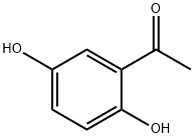
The second alternative method first employs benzyl groups to protect the hydroxyl functional group in 2,5-dihydroxyacetophenone, followed by an O-alkylation reaction under the same conditions to give acetophenone derivatives (1-6) in high yields (95%). Next, the acetophenone derivatives (1-6) were converted by the Vilsmeier-Haack reaction under standard conditions (70% yield) to the products (1-7) which were previously synthesized by other methods. Finally, the product (1-7) was subjected to Knoevenagel condensation reaction with diethyl malonate to give the target diester (1-8) in high yield (94%). Based on the analysis of the results of Scheme 2, it can be observed that both methods can be used for the synthesis of intermediates (1-7) from 2,5-dihydroxyacetophenone (1-4) in overall yields of 42% and 66%, respectively. However, the second method showed superior performance in terms of yield.
[1] Patent: WO2018/156459, 2018, A1. Location in patent: Page/Page column 22; 23
[2] Bioorganic and Medicinal Chemistry, 2010, vol. 18, # 1, p. 249 - 266
[3] Patent: US2007/88160, 2007, A1. Location in patent: Page/Page column 10-11
[4] Patent: US2010/22770, 2010, A1. Location in patent: Page/Page column 5-6
[5] Bioorganic and Medicinal Chemistry Letters, 2010, vol. 20, # 4, p. 1410 - 1414