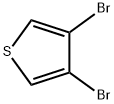
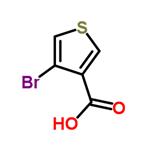
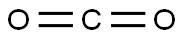Cool 100 mL of ether to -78°C under a nitrogen atmosphere. A 1.6 M n-butyllithium solution (28.4 mL) was slowly added. Subsequently, a solution of 3,4-dibromothiophene (10 g, 41.3 mmol) dissolved in 50 mL of ether was added dropwise over 10 min. The reaction temperature was maintained at -78 °C and stirring was continued for 10 min before an excess (>50 g) of fresh powdered dry ice (CO) was added. The reaction mixture was stirred at -78°C for 1 hour, then 30 mL of 1 M NaOH solution (pre-diluted with 100 mL of water) was slowly added (note: CO will be released). Gradually increase the temperature until the ice melts. Separate the organic and aqueous phases and extract the ether phase with 25 mL of 1 N NaOH solution. Combine all aqueous phases and acidify with 100 mL of 1 N HCl solution to pH < 7. Collect the precipitated white precipitate by filtration, wash with water, and dry in a vacuum oven to give 4-bromothiophene-3-carboxylic acid as a white solid (5.8 g, 68% yield).
[1] Pharmazie, 2006, vol. 61, # 11, p. 901 - 907
[2] Journal of the Chemical Society, Perkin Transactions 1: Organic and Bio-Organic Chemistry (1972-1999), 1994, # 19, p. 2735 - 2744
[3] Patent: WO2006/3096, 2006, A1. Location in patent: Page/Page column 71-72
[4] Patent: US2013/116233, 2013, A1. Location in patent: Paragraph 1018; 1019
[5] Patent: WO2014/140086, 2014, A1. Location in patent: Page/Page column 68-69