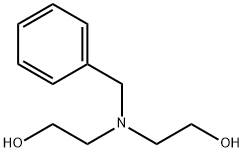
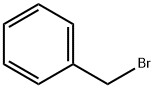
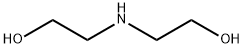
General procedure for the synthesis of 2,2-(benzylimino)diethanol from benzyl bromide and diethanolamine: K2CO3 (8.65 g, 62.58 mmol) and benzyl bromide (2.96 g, 25 mmol) were added to anhydrous acetone (65 mL) followed by diethanolamine (2.63 g, 53.08 mmol). The reaction mixture was heated to reflux and the reaction was continued overnight. After completion of the reaction, the white suspension obtained was filtered and the solvent was removed by evaporation to obtain 5.6 g of yellow liquid. The liquid was dissolved in dichloromethane (DCM) and washed with deionized water. The organic layer was dried over anhydrous sodium sulfate and concentrated in vacuum to give the final 2,2-(benzylimino)diethanol (4.88 g, 100% yield). The product was analyzed by HPLC-MS (Method A): retention time 1.14 min; ESI-MS m/z 196 ([M+H]+).
[1] Inorganic Chemistry, 2012, vol. 51, # 19, p. 10384 - 10393,10
[2] Patent: WO2017/16669, 2017, A1. Location in patent: Page/Page column 201
[3] Journal of Medicinal Chemistry, 2003, vol. 46, # 12, p. 2376 - 2396
[4] Patent: EP970046, 2003, B1. Location in patent: Page 45-46
[5] Patent: US6342508, 2002, B1. Location in patent: Page column 58-59