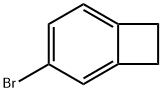
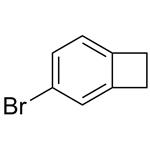
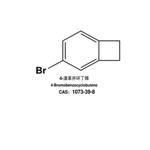
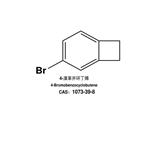
4-Bromobenzocyclobutene is used in organic synthesis. It is also used in the production of pharmaceuticals, agrochemicals, and other fine chemicals.
The bromine atom in 4-bromobenzocyclobutene is highly reactive and can undergo various chemical reactions, such as substitution, elimination, and coupling reactions.
The general procedure for the synthesis of 4-bromobenzocyclobutene from benzocyclobutene is as follows: first, α-chloro-o-xylene 1 is subjected to a pyrolysis reaction at about 800 °C and 0.5 mbar to produce benzocyclobutene 2 in 45% yield. Subsequently, benzocyclobutene 2 was dissolved in acetic acid and reacted with a mixture of bromine and iodine at room temperature for selective bromination to give 4-bromobenzocyclobutene 3. 3 was dissolved in toluene and a slightly molar excess of 1,4-dihydro-1,4-epoxynaphthalene 4 was added and reacted for 20 hr at 220° C. to give the Diels-Alder addition product 5 as a pure inner/outer in 80% yield mixture, which was a colorless crystalline substance. -yl)-5,12-dihydroanthracene 7. After recrystallization of compound 7 with o-dichlorobenzene, the dehydrogenation reaction was carried out by treatment with 2,3-dichloro-5,6-dicyano-p-benzoquinone (DDQ) in boiling o-xylene. After repeated vacuum sublimation purification, orange-red crystals of 2-(anthracen-2-yl)tetraphenyl 8 were finally obtained in 75% yield. All intermediates were characterized by 1H NMR, 13C NMR spectroscopy and mass spectrometry. Compound 8 was characterized by UV-visible spectroscopy.
[1] Chemistry - A European Journal, 2017, vol. 23, # 54, p. 13445 - 1345
[2] Patent: US2008/214838, 2008, A1. Location in patent: Page/Page column 2-3
[3] Tetrahedron, 1965, vol. 21, p. 245 - 254
[4] Journal of the Chemical Society [Section] B: Physical Organic, 1969, p. 12 - 15
[5] Patent: EP2236500, 2010, A1. Location in patent: Page/Page column 21