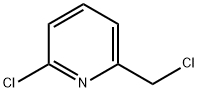
The general procedure for the synthesis of 2-chloro-6-(chloromethyl)pyridine from 2-chloro-6-hydroxymethylpyridine is as follows:
Preparation of Example 52-1-2 2-chloro-6-(chloromethyl)pyridine: To a mixed solution containing (6-chloropyridin-2-yl)methanol (200 mg, 1.39 mmol) and toluene (3 mL), thionyl chloride (152 μL, 2.09 mmol) was slowly added under ice bath conditions. The reaction mixture was then stirred for 2 h at room temperature. Upon completion of the reaction, saturated aqueous sodium bicarbonate solution was added to the mixture to neutralize the excess thionyl chloride, followed by extraction with ethyl acetate. The organic layer was separated, washed sequentially with water and saturated aqueous sodium chloride solution, dried over anhydrous magnesium sulfate, and filtered. The filtrate was concentrated under reduced pressure and the residue was purified by silica gel column chromatography (eluent: heptane/ethyl acetate=4:1) to afford the target product 2-chloro-6-(chloromethyl)pyridine (163.2 mg, 73% yield).
1H-NMR (CDCl3) δ (ppm): 4.64 (2H, s), 7.29 (1H, d, J=8.0 Hz), 7.44 (1H, d, J=7.6 Hz), 7.70 (1H, dd, J=7.6,8.0 Hz).