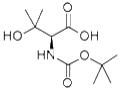
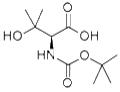
The general procedure for the synthesis of (S)-2-((tert-butoxycarbonyl)amino)-3-hydroxy-3-methylbutanoic acid from tert-butyl (R)-(1,3-dihydroxy-3-methylbutan-2-yl)carbamate was as follows: 2,2,6,6-tetramethylpiperidinium 1-yloxy (TEMPO, 2.3 g, 15 mmol) was added to (R)-(1,3-dihydroxy-3-methyl tert-butyl (33.0 g, 150 mmol) carbamate in acetonitrile (750 mL) and sodium phosphate buffer (600 mL, 0.7 M, pH 6-7) and heated to 35 °C. Subsequently, sodium chlorite solution (34.2 g, dissolved in 150 mL of water) and dilute sodium hypochlorite solution (3 mL of commercial solution diluted in 100 mL of water, 60 drops total) were added dropwise simultaneously to the reaction mixture. The reaction mixture was stirred at 35°C overnight, cooled to room temperature, then the pH was adjusted to 3 with citric acid (~15 g), saturated with sodium chloride, and extracted with ethyl acetate (3 x 2 L). The organic phases were combined and concentrated under reduced pressure. The residue was dissolved in 1.5 L of 2 M sodium carbonate solution and washed with ethyl acetate (2 x 2 L). The aqueous layer was cooled to 0°C, the pH was adjusted to 3.0 with 2M phosphoric acid solution and saturated again with sodium chloride. Extracted with ethyl acetate (3×2L), the organic phases were combined, dried, filtered and concentrated under reduced pressure to give (S)-2-((tert-butoxycarbonyl)amino)-3-hydroxy-3-methylbutanoic acid (28.4 g, 81% yield) as a colorless solid. NMR (400 MHz, DMSO-d6): δ= 1.15 (s, 3H), 1.17 (s, 3H), 1.39 (s, 9H), 3.86 (d, J = 8.6 Hz, 1H), 6.52 (d, J = 8.9 Hz, 1H).
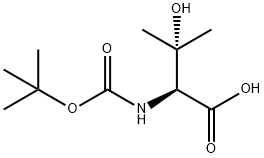
![Carbamic acid, [(1R)-2-hydroxy-1-(hydroxymethyl)-2-methylpropyl]-, 1,1-](/CAS/20180713/GIF/473545-40-3.gif)