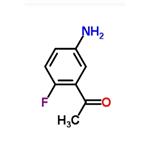
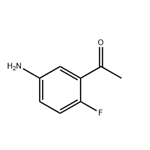
General procedure for the synthesis of 2-fluoro-5-aminoacetophenone from 1-(2-fluoro-5-nitrophenyl)ethanone: 1-(2-fluoro-5-nitrophenyl)ethanone (0.3 g, 1.64 mmol), iron powder (0.27 g, 4.92 mmol), ammonium chloride (0.17 g, 3.28 mmol), isopropanol (12.8 mL), and water (3.2 mL ) were mixed and reacted for 3 hours. After completion of the reaction, the insoluble precipitate was removed by filtration, the organic layer was extracted with dichloromethane, dried with anhydrous magnesium sulfate, and concentrated in vacuum to obtain a yellow oily crude product. The crude product was purified by silica gel column chromatography (eluent: ethyl acetate/hexane=1:2, Rf=0.3) to afford 2-fluoro-5-aminoacetophenone (0.19 g, 76% yield).
[1] Journal of Heterocyclic Chemistry, 1991, vol. 28, # 3, p. 673 - 683
[2] Patent: WO2016/172255, 2016, A1. Location in patent: Page/Page column 96; 97
[3] Bioorganic and Medicinal Chemistry Letters, 2003, vol. 13, # 10, p. 1725 - 1728
[4] Patent: EP2147914, 2010, A1. Location in patent: Page/Page column 29
[5] Patent: US2012/15961, 2012, A1. Location in patent: Page/Page column 41