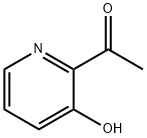
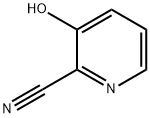
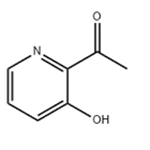
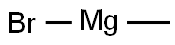General procedure for the synthesis of 1-(3-hydroxypyridin-2-yl)ethanone from methylmagnesium bromide and 2-cyano-3-hydroxypyridine: Referring to the method of Example 58, to a solution of tetrahydrofuran (THF, 50 mL) of 2-cyano-3-hydroxypyridine (3.00 g, 25.0 mmol) was added slowly, under cooling in an ice bath, a THF solution of 3M methylmagnesium bromide ( 25 mL). After addition, the reaction mixture was stirred at room temperature for 30 min. Subsequently, the reaction mixture was neutralized with 6N hydrochloric acid solution and extracted twice with ethyl acetate. The organic layers were combined, washed with saturated sodium chloride solution and dried over anhydrous sodium sulfate. The solvent was removed by concentration under reduced pressure to afford the title compound 1-(3-hydroxypyridin-2-yl)ethanone (3.43 g, quantitative yield) as a powdered solid. The crude product was used directly in the subsequent reaction without further purification.