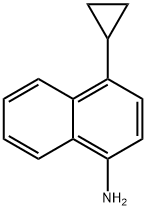
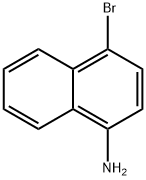
Tetrakis(triphenylphosphine)palladium (0.26 g, 0.23 mmol) and potassium phosphate (1.67 g, 7.87 mmol) were added to a 50 mL round-bottomed flask under nitrogen protection using 4-bromo-1-naphthylamine (0.50 g, 2.25 mmol) and cyclopropylboronic acid (0.212 g, 2.47 mmol). A solvent mixture of dioxane and water (25:1, 26 mL) was then added, and after three nitrogen displacements, the reaction mixture was heated to reflux in a 90 °C oil bath for 6 hours. After completion of the reaction, it was cooled to room temperature and filtered to remove insoluble material. The filtrate was concentrated under reduced pressure to remove the solvent, diluted with 50 mL of water and extracted with ethyl acetate (2 x 10 mL). The organic layers were combined, washed twice with saturated brine, dried over anhydrous sodium sulfate, filtered and concentrated to give the intermediate 4-cyclopropyl-1-aminonaphthalene as a purplish red oil in 95.3% yield.