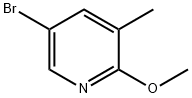
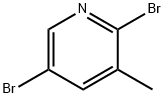
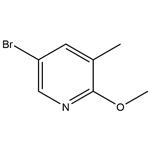
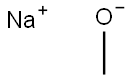A) Synthesis of 5-bromo-2-methoxy-3-methylpyridine. 2,5-Dibromo-3-methylpyridine (2.08 g, 8.3 mmol) was suspended in a methanolic solution (17 mL) of 2 M sodium methanol and heated by single mode microwave radiation at 120 °C for 40 min. After completion of the reaction, the reaction mixture was slowly poured into a mixture of ice and 1 M aqueous hydrochloric acid solution and extracted twice with dichloromethane. The organic layers were combined, dried with anhydrous sodium sulfate, filtered and concentrated under reduced pressure to afford the crude product 5-bromo-2-methoxy-3-methylpyridine 1.57 g (89% yield), which could be used in the subsequent reaction without further purification. The product was characterized by 1H NMR (400 MHz, with chloroform-d as solvent and internal standard): δ 8.02 (d, 1H, J = 2.3 Hz), 7.45-7.47 (m, 1H), 3.92 (s, 3H), 2.16 (broad single peak, 3H).