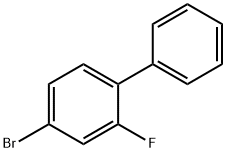
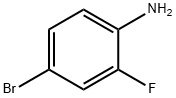
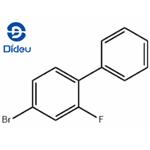
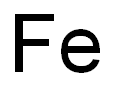General procedure for the synthesis of 4-bromo-2-fluorobiphenyl from 4-bromo-2-fluoroaniline and iron powder: (1) A solution of 96 g (0.50 mol) of crude 4-bromo-2-fluoroaniline and 60.0 g (1.0 mol) of glacial acetic acid in 100 ml of benzene was added dropwise over a period of 7 hours to a mixture of 69.0 g (1.0 mol) of sodium nitrite, 69 ml of water and 700 ml of benzene. The temperature was maintained at 65°C. Subsequently, the reaction mixture was stirred at 65°C for 12 hours under nitrogen protection. Upon completion of the reaction, the cooled mixture was washed twice with 400 mL of 1N hydrochloric acid. Next, 20 g (0.36 mol) of iron powder, 250 mL of methanol and 150 mL (1.8 mol) of concentrated hydrochloric acid were added and heated at reflux for 13 hours. After the reaction solution was cooled, the benzene layer was washed with 490 ml of water and the solvent was evaporated under reduced pressure at 40 °C/40 mmHg. Finally, the dark-colored oily material obtained was distilled at a pressure of 10 mmHg and the fractions with boiling points of 132-141 °C/8 mmHg were collected to give 64.6 g (51.5% yield) of 4-bromo-2-fluorobiphenyl. The product crystallized after inoculation with crystal seed. A variant of the sodium nitrite method was used to synthesize 4-bromo-2-fluorobiphenyl IV1 and the corresponding results were obtained as shown in Table I.