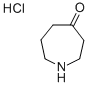
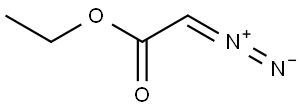
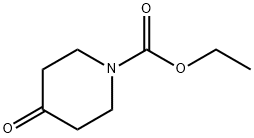
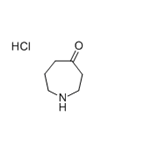
Step 1: Synthesis of azepan-4-one hydrochloride (intermediate): To a stirred solution of N-ethoxycarbonyl-4-piperidone (600 mg, 3.00 mmol) in anhydrous ether (1.5 mL) under nitrogen protection was added simultaneously a solution of boron trifluoride in ethyl ether (380 μL, 3.00 mmol) and ethyl diazoacetate (412 μL, 3.92 mmol) each dissolved in anhydrous ether (0.4 mL) to a solution at a reaction temperature of -35 °C. The reaction mixture was stirred continuously at -35 °C for 1 h 30 min, followed by slow warming to room temperature. After completion of the reaction, the reaction solution was washed with 30% aqueous potassium carbonate (3 mL) and the organic phase was extracted with ethyl acetate (3 x 5 mL). The organic layers were combined, dried with anhydrous sodium sulfate, filtered and concentrated under reduced pressure. The resulting crude product was dissolved in 4N aqueous hydrochloric acid (13 mL) and refluxed for 6 hours. At the end of the reaction, the solvent was removed under reduced pressure to give a light yellow oil (400 mg, 88% yield). The product was characterized by 1H NMR (400 MHz, CDCl3): δ 3.04 (m, 4H, CH2NH), 2.59 (m, 4H, CH2CO), 1.86 (m, 1H, NH), 1.75 (m, 2H, CH2CH2NH).