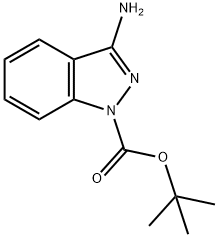
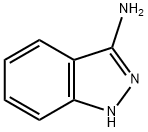
General procedure for the synthesis of tert-butyl 3-amino-1H-imidazole-1-carboxylate from di-tert-butyl dicarbonate and 3-aminoindazole: first, 150 mg (1.13 mmol) of 1H-indazol-3-amine was dissolved in 3 mL of THF, followed by the addition of 320 mg (1.46 mmol) of di-tert-butyl dicarbonate, 137 mg (1.35 mmol) of triethylamine, and 48 mg (0.39 mmol) of 4-dimethylaminopyridine. The reaction mixture was stirred at room temperature for 1.5 hours. After completion of the reaction, the reaction solution was diluted with ethyl acetate and washed sequentially with water, saturated aqueous ammonium chloride solution and saturated sodium chloride solution each. The organic phase was dried with anhydrous sodium sulfate, filtered and the filtrate was concentrated. The residue was purified by silica gel column chromatography (eluent: cyclohexane/ethyl acetate, 3:1→1:1) to give 126 mg of tert-butyl 3-amino-1H-imidazole-1-carboxylate in 48% yield.LC-MS (Method 2): Rt = 0.88 min; MS (ESpos): m/z = 234 (M + H)+; 1H-NMR (400 MHz. DMSO-d6): δ = 1.58 (s, 9H), 6.30 (s, 2H), 7.25 (t, 1H), 7.50 (t, 1H), 7.82 (d, 1H), 7.94 (d, 1H).