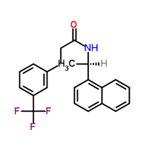
In another synthetic scheme, the reduction of the double bond and amide carbonyl group of (E)-N-((R)-1-naphthalen-1-yl-ethyl)-3-(3-trifluoromethylphenyl)acrylamide is carried out in a stepwise manner. Optionally, either the carbonyl or the olefinic double bond is reduced first. In the case of the reduction of the olefinic double bond, the reaction is realized by hydrogenation in the presence of a metal catalyst. This was done as follows: (E)-N-((R)-1-naphthalen-1-yl-ethyl)-3-(3-trifluoromethylphenyl)acrylamide (1.0 equiv; 0.67 mmol; 250 mg) was suspended with 10 wt% Pd/C (25 mg) in a solvent mixture of methanol and toluene (7 mL/1 mL). The reaction flask was evacuated and backfilled with a hydrogen balloon (repeated three times). The reaction mixture was stirred at room temperature and the progress of the reaction was monitored by high performance liquid chromatography (HPLC).After 90 min, the reaction mixture was filtered through a bed of diatomaceous earth and the solvent in the filtrate was removed under vacuum. The resulting solid was dried in a vacuum oven at 45 °C overnight. The isolated yield was 91.2% (229 mg). The structure of the product was confirmed by 1H NMR and mass spectrometry.1H NMR (400 MHz, CDCl3) δ ppm: 1.6 (d, 3H), 2.39-2.53 (m, 2H), 2.96-3.13 (m, 2H), 5.55 (br.s, 1H), 5.87-5.96 (m, 1H), 7.29-7.38 (m, 2H), 7.39-7.46 (m, 2H), 7.29-7.38 (m, 2H), 7.39-7.38 (m, 2H). 7.39-7.46 (m, 4H), 7.46-7.55 (m, 2H), 7.76-7.82 (m, 1H), 7.83-7.89 (m, 1H), 8.00-8.07 (m, 1H). High Resolution Mass Spectrometry (HRMS) (M+1): 372.1.
![2-PropenaMide, N-[(1R)-1-(1-naphthalenyl)ethyl]-3-[3-(trifluoroMethyl)phenyl]-, (2E)-](/CAS/20150408/GIF/1095393-66-0.gif)