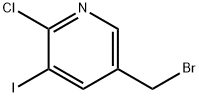
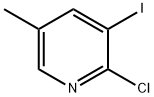
General procedure for the synthesis of 5-(bromomethyl)-2-chloro-3-iodopyridine from 2-chloro-3-iodo-5-methylpyridine: 4.60 g (18.15 mmol) of 6-chloro-5-iodo-3-methylpyridine (Setliff et al., J. Chem. Engineering Data (1976), 21(2), 246-7), 3.39 g (19.06 mmol) of N-bromosuccinimide and 0.30 g (1.82 mmol) of 2,2'-azobis(2-methylpropionitrile) were dissolved in 500 ml of chlorobenzene and reacted under reflux conditions for about 16 hours. Upon completion of the reaction, the reaction mixture was washed with saturated aqueous sodium sulfite solution and sodium bicarbonate solution, followed by drying with sodium sulfate and concentration under reduced pressure. Purification by silica gel column chromatography (silica gel 60-Merck, particle size: 0.04 to 0.063 mm) with the mobile phase ethyl acetate: cyclohexane (1:10) afforded 3.86 g (38% yield) of the target product 3-bromomethyl-6-chloro-5-iodopyridine.1H-NMR (CD3CN) data: δ [ppm] = 4.48 (s, 2H), 8.30 ( s, 1H), 8.40 (s, 1H).