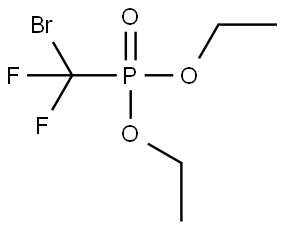
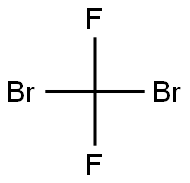
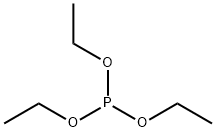
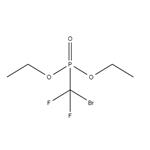
Dibromodifluoromethane (209.8 g, 1 mol) was slowly added dropwise to a solution of triethyl phosphite (153.5 g, 0.925 mol) in anhydrous ether (450 mL) equipped with a magnetic stirrer under nitrogen protection. After the dropwise addition, the reaction system was slowly warmed to room temperature and subsequently heated to reflux for 20 hours. Upon completion of the reaction, the volatile solvent was removed by distillation under reduced pressure and the fraction with a boiling point of 50-52 °C (1 mmHg) was collected to afford the colorless liquid product diethyl (bromodifluoromethyl) phosphonate (256.3 g, 96% yield). The structure of the product was confirmed by 1H NMR (CDCl3): δ 4.35 (m, 4H), 1.35 (m, 6H); 13C NMR (CDCl3): δ 122.9, 119.8, 118.6, 115.4, 114.2, 111.1, 66.59, 66.50, 16.56, 16.48.