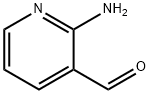
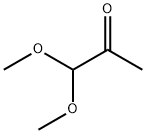
The general procedure for the synthesis of [1,8]naphthyridine-2-carboxaldehyde dimethyl acetal from 2-amino-3-pyridinecarboxaldehyde and 1,1-dimethoxyacetone was as follows: to a mixed solution of 2-amino-3-formylpyridine (40 g, 0.316 mol), ethanol (267 ml) and water (41 ml) at 6 °C, acetone aldehyde dimethyl acetal (51.3 ml ) while controlling the rate of addition to keep the internal temperature below 20°C. Subsequently, 5M NaOH solution (82.3 ml, 0.411 mol) was added. The reaction mixture was stirred at room temperature for 1 hour before the ethanol was removed under reduced pressure. Isopropyl acetate (iPAc, 100 mL) and sodium chloride (55 g) were added to the residue to separate the organic and aqueous layers. The aqueous layer was extracted with isopropyl acetate (2 x 100 mL). All organic layers were combined, filtered through a silica gel bed (90 g) and rinsed with isopropyl acetate (1 L). The filtrate and rinse solution were combined and concentrated to 200 ml at 38 °C. Hexane (400 ml) was slowly added to the concentrate and the resulting suspension was cooled to 10 °C and aged for 30 min, then filtered. The filter cake was dried under reduced pressure to give the colorless crystalline product [1,8]naphthyridine-2-carbaldehyde dimethanol (54.2 g, 84% yield) with a melting point of 53.5-55.5 °C. Addition of additional hexane (100 mL) to the mother liquor afforded a further 7.2 g (11% yield) of product after filtration. The NMR hydrogen spectrum (300 MHz, CDCl3) data of the product were as follows: δ 8.89 (dd, J = 4.3 and 2.0 Hz, 1H), 8.03 (d, J = 8.4 Hz, 1H), 7.98 (dd, J = 8.1 and 2.0 Hz, 1H), 7.56 (d, J = 8.4 Hz, 1H), 7.26 (dd, J = 8.1 and 4.3 Hz, 1H), 5.28 (s, 1H), 3.30 (s, 6H). The NMR carbon spectrum (75.5 MHz, CDCl3) data are as follows: δ 161.3, 155.0, 153.5, 137.9, 136.8, 122.5, 122.3, 119.4, 105.9, 54.9.
![2-DIMETHOXYMETHYL-[1,8]NAPHTHYRIDINE Structure](/CAS/GIF/204452-90-4.gif)