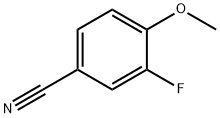
Example 2.1.1: Preparation of (3-fluoro-4-methoxyphenyl) methylamine (ICb). To a solution of 3-fluoro-4-methoxybenzyl cyanide (ICa, 68.0 g, 450 mmol, 1 equiv) in methanol (2 L) was added Raney nickel (Raney-Ni, 70.0 g). After addition, the reaction mixture was stirred at room temperature overnight. After completion of the reaction, the solid formed was removed by filtration and the solid was washed with methanol. The clarified filtrate was concentrated under reduced pressure to afford the title compound (3-fluoro-4-methoxyphenyl)methylamine (ICb, 62.8 g, 90% yield) as a yellow oil.