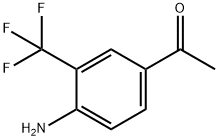
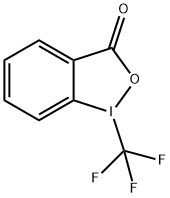
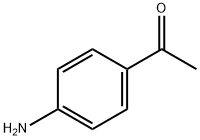
1-(Trifluoromethyl)-1,2-phenyliodono-3(1H)-one (0.4 mmol), 4-aminoacetophenone (0.2 mmol), and Ir(ppy)3 (2 mg) were placed in a reaction flask containing 1 mL of DMF under nitrogen or argon protection. The reaction mixture was irradiated with a blue LED strip (7 W) at room temperature until the trivalent iodine reagent was completely converted. Upon completion of the reaction, 10 mL of saturated aqueous Na2CO3 solution was added and extracted three times with ethyl acetate. The organic layers were combined, washed sequentially with water and saturated brine, and then dried with anhydrous Na2SO4. The dried organic layer was purified by column chromatography (eluent ratio: petroleum ether 60-90:ethyl acetate=15:1 to 8:1) to afford the target product 1-(4-amino-3-(trifluoromethyl)phenyl)ethanone in 61% yield.
[1] Patent: CN103553857, 2016, B. Location in patent: Paragraph 0025-0026
[2] Organic Letters, 2014, vol. 16, # 6, p. 1768 - 1771
[3] Organic Letters, 2018, vol. 20, # 13, p. 3732 - 3735
[4] Patent: CN108503552, 2018, A. Location in patent: Paragraph 0150-0154