General method C: Preparation of compounds A17-A24 by catalytic Wittig reaction. catalyst Aid (0.2 mmol, 20 mol%) and base A2 (2.0-3.5 mmol, 2.0-3.5 eq.) were added to a 1-dram vial fitted with a stirrer in air. If a solid aldehyde (1.0-1.2 mmol, 1.0-1.2 eq.) is used, it is added together at this point. The vial is then sealed with a septum and the air in the vial is replaced with argon. Toluene (1.4 mL) and the liquid reagents: aldehydes (1.2 mmol, 1.2 eq.) and organic halides (1.0 mmol, 1.0 eq.) were added sequentially. Diphenylsilane (1.2 mmol, 1.2 eq.) was then added and the septum was replaced with a PTFE-lined screw cap under an inert atmosphere. The reaction mixture was heated at 140°C or 150°C for 24-48 hours. After the reaction has been carried out for 24 hours, the base and halide are added additionally and the reaction is continued up to 48 hours. After completion of the reaction, the crude product was filtered through Celite, concentrated under reduced pressure and finally purified by fast column chromatography.
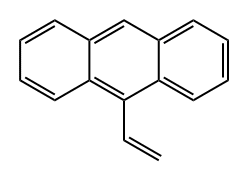
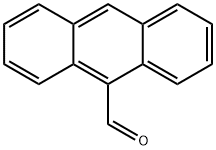
Synthesis of 9-vinylanthracene (A23): In toluene (0.7 mL), 9-anthracenecarboxaldehyde (128 mg, 0.6 mmol, 1.2 eq.), iodomethane (31 μL, 0.5 mmol, 1.0 eq.), and diphenylsilane (112 μL, 0.6 mmol, 1.2 eq.) were added to the reaction with catalyst A3b (30 mg, 20 mol%) and base A2 (210 mg, 1.5 mmol, 3.0 eq.) were added, and the reaction was carried out at 140 °C for 24 hours. During the reaction, iodomethane and A2 were added twice at 0 h and 4 h. The crude product was purified by fast column chromatography (eluent: hexane, Rf = 0.35) to afford 9-vinylanthracene (A23, 66 mg, 65% yield) in yellow liquid form.1H NMR (400 MHz, CDCl3) δ: 5.68 (d, J = 18.0 Hz, 1H), 6.06 (d, J = 18.0 Hz, 1H), 6.06 (d, J = 18.0 Hz, 1H) 6.06 (d, J = 11.5 Hz, 1H), 7.48-7.60 (m, 5H), 8.00-8.08 (m, 2H), 8.35-8.40 (m, 2H).