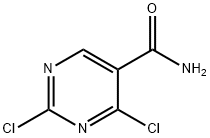
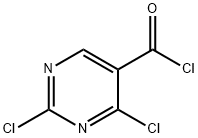
5.1.32 Synthesis of 2,4-dichloropyrimidine-5-carboxamide (27): 2,4-dihydroxypyrimidine-5-carboxylic acid (25) (2.98 g, 19.1 mmol) was dissolved in POCl3 (7.10 mL, 76.4 mmol) followed by addition of PCl5 (13.1 g, 63.0 mmol). The reaction mixture was heated to reflux for 4.5 hours. After completion of the reaction, the mixture was cooled and concentrated under vacuum. Toluene (20 mL) was added to the concentrated residue and again concentrated under vacuum. The residue was dissolved in CH2Cl2 (20 mL) and filtered. The filtrate was concentrated under vacuum to afford 2,4-dichloropyrimidine-5-carbonyl chloride (26) as a yellow oil (4.38 g), which could be used in the next step without further purification. A solution of CH2Cl2 (5 mL) of 26 (4.38 g) was slowly added to a mixture of 28% aqueous NH3 (3 mL) and H2O (3 mL) at -10 °C and the reaction was stirred for 5 min keeping the temperature at -10 °C. At the end of the reaction, the organic solvent was removed under reduced pressure and the precipitate was collected by filtration to afford the target product 2,4-dichloropyrimidine-5-carboxamide (27) (3.13 g, 16.3 mmol, 85% yield) as a light yellow solid. The product was confirmed by 1H NMR (DMSO-d6) δ: 8.05 (1H, s), 8.17 (1H, s), 8.90 (1H, s) and MS (ESI) m/z: 192 (M + H)+.