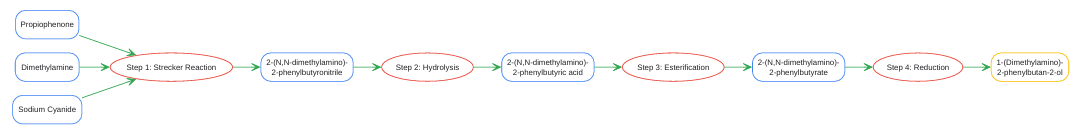
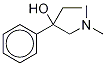1-(Dimethylamino)-2-phenylbutan-2-ol is primarily synthesised via a Strecker synthesis-based route. Strecker Synthesis-Based Route: This multi-step synthesis begins with the reaction of propiophenone, dimethylamine, and a cyanide source (like sodium cyanide) to form the α aminonitrile, 2-(N,N-dimethylamino)-2-phenylbutyronitrile. This intermediate then undergoes hydrolysis to the corresponding carboxylic acid, followed by esterification and finally reduction of the ester to yield the target amino alcohol.