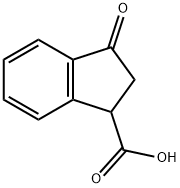
3-Oxo-1-indancarboxylic acid participates in the synthesis of 3-hydroxymethyl-1-indanol (±). Enantiomeric separation of 3-oxo-1-indancarboxylic acid on liquid chromatography chiral stationary phases (CSPs) by supercritical fluid chromatography (SFC) has been reported. Separation of 3-oxo-1-indancarboxylic acid enantiomers using a new immobilized polysaccharide chiral stationary phase, CHIRALPAK IA with hexane has been described.
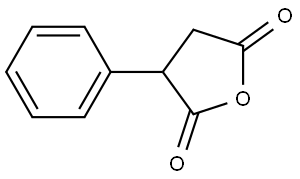
The general procedure for the synthesis of 3-carbonyl-1-indenoic acid from phenylsuccinic anhydride was as follows: phenylsuccinic anhydride (10 g, 57 mmol) was dissolved in 1,2-dichloroethane (200 mL). The solution was slowly added dropwise to a solution of 1,2-dichloroethane (50 mL) containing aluminum trichloride (17 g, 130 mmol) at 0°C. After the dropwise addition was completed, the reaction mixture was continued to be stirred for 1 hour at room temperature. Subsequently, water (50 mL) was added at 0 °C to terminate the reaction. The reaction mixture was extracted with ethyl acetate (3 x 60 mL), the organic phases were combined and concentrated by evaporation. After drying, the light yellow oily product 5A (8.3 g, 83% yield) was obtained.
[1] Bulletin de la Societe Chimique de France, 1982, vol. 2, # 5-6, p. 161 - 166
[2] Patent: CN108250128, 2018, A. Location in patent: Paragraph 0195; 0197; 0198; 0199
[3] Patent: WO2016/44770, 2016, A1. Location in patent: Page/Page column 661
[4] Tetrahedron, 2004, vol. 60, # 41, p. 9245 - 9253
[5] Canadian Journal of Chemistry, 1961, vol. 39, p. 2563 - 2571