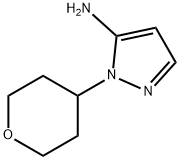
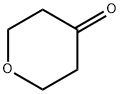
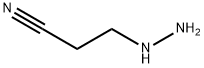
3-Hydrazinylpropionitrile (3.40 g, 40 mmol) was added slowly and dropwise to a solution of tetrahydropyran-4-one (4.00 g, 40 mmol) in ethanol (40 mL) at room temperature. The reaction mixture was stirred at room temperature for 3 hours. Subsequently, the mixture was concentrated under reduced pressure and the residue was dissolved in n-butanol (80 mL) and sodium methanolate (4.32 g, 80 mmol) was added. The mixture was refluxed overnight. After the reaction was complete, the mixture was concentrated under reduced pressure. The residue was treated with a solution of saturated sodium bicarbonate and ammonium chloride (80 mL). The aqueous phase was extracted with dichloromethane (60 mL × 2) and the combined organic phases were dried with anhydrous sodium sulfate. The organic phase was concentrated and the residue was purified by fast column chromatography (acetonitrile/water = 30%) to afford 5.34 g of crude 1-(tetrahydro-2H-pyran-4-yl)-1H-pyrazol-5-amine as a yellow solid. Mass spectrometry analysis showed m/z 168.3 ([M + H]+).