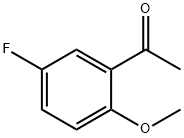
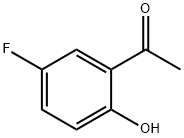
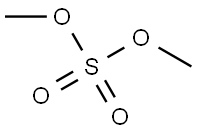
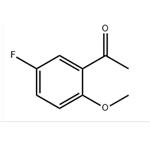
Step 2. A 4.1:1 mixture of 5-fluoro-2-hydroxyacetophenone (10.4 g, 61.8 mmol) and dimethyl sulfate (2.54 g, 16.5 mmol) was dissolved in acetone (50 mL) and potassium carbonate (2.50 g, 18.1 mmol) was added. Dimethyl sulfate (0.25 mL, 2.6 mmol) was then added. The reaction mixture was heated to reflux for 18 h. After cooling to room temperature, water (20 mL) was added. The mixture was stirred for 3 hours at room temperature and then partitioned between dichloromethane (50 mL) and brine (50 mL). The organic layer was separated and the aqueous layer was extracted with dichloromethane (3 x 50 mL). The organic phases were combined, dried over anhydrous sodium sulfate and concentrated under reduced pressure. 13.0 g (98% yield, 97% area) of 2-methoxy-5-fluoroacetophenone was obtained as a yellow oil.NMR (400 MHz, CDCl3) δppm: 2.64 (s, 3H), 3.92 (s, 3H), 6.94 (dd, J=9.09,4.04 Hz, 1H), 7.18 (ddd, J=9.09, 7.33,3.28 Hz, 1H), 7.48 (dd, J=8.97,3.16 Hz, 1H); HPLC retention time: 3.39 min; MS (ESI+) C9H9FO2 m/z 169.1 (M+H)+.