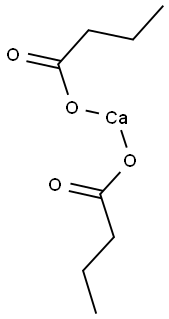
Room temperature stirring, butyric acid 88.11g (1.0mol, 1.0eq) in 5 batches dissolved in 200mL of aqueous solution containing 45.0g sodium hydroxide, and then to the reaction solution slowly added 200mL of aqueous solution containing calcium chloride 108.5g (0.5mol, 0.5eq), and gradually there is a white solid precipitation, the feeding of the reaction solution to continue to stir the reaction for 1h, heating and temperature rise The reaction solution was heated to 70 and continued to react for 0.5h, filtered while hot, the cake was washed with hot water until the filtrate was neutral, dried at 50 under reduced pressure to obtain calcium butyrate 155.77g, yield 72.7%.
Recrystallise it from water (5mL/g) by partial evaporation in a desiccator and dry it in a vacuum to constant weight. [Pathak & Bhide J Indian Chem Soc 30 47, 48 1953.] Its dissociation constant at 25o is 0.29 [Colman-Porter & Monk J Chem Soc 4363 1952, Beilstein 2 IV 785].