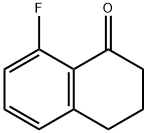
Step C: Preparation of 8-fluoro-3,4-dihydronaphthalen-1(2H)-one
8-Amino-3,4-dihydronaphthalen-1(2H)-one (9.1 g, 56.5 mmol) was dissolved in dichloromethane (415 mL) at 0 °C and boron trifluoride ether compound (12.0 g, 84.7 mmol) was added. After stirring the mixture for 10 minutes, a solution of tert-butyl nitrite (7.04 g, 68.3 mmol) in dichloromethane (50 mL) was added dropwise. The reaction was stirred vigorously at 0°C for 1 hour. The solution was then cooled in a dry ice bath, diluted with pentane (415 mL) and stirred for 10 minutes. Stirring was stopped and after the solid settled, the solvent was removed. This operation was repeated once and the resulting solid was vacuum dried. The dried solid was heated in heptane at 100 °C for 2 hours. After the reaction was cooled to room temperature, the product was dissolved in dichloromethane and washed sequentially with water and brine. The organic layers were combined, dried over anhydrous magnesium sulfate, filtered and concentrated. The residue was purified by Biotage column chromatography (silica gel, hexane/ethyl acetate gradient) to afford 8-fluoro-3,4-dihydronaphthalen-1(2H)-one (4.92 g, 53% yield).
LCMS m/z = 165.2 [M + 1]+.
1H NMR (400 MHz, CDCl3) δ 2.09-2.13 (m, 2H), 2.64-2.67 (m, 2H), 2.97 (t, J = 12.2 Hz, 2H), 6.95-6.99 (m, 1H), 7.04 (d, J = 7.6 Hz, 1H), 7.38-7.43 (m, 1H).