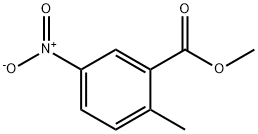
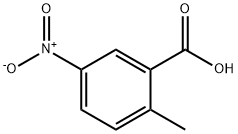
4.2.1 Synthesis of methyl 2-methyl-5-nitrobenzoate (2): 2-methyl-5-nitrobenzoic acid (15 g, 82.8 mmol) and potassium carbonate (K2CO3, 17.2 g, 124.2 mmol) were suspended in N,N-dimethylformamide (DMF, 75 mL) in a dry round-bottom flask. To this suspension, iodomethane (6.7 mL) was slowly added dropwise at room temperature. The reaction mixture was stirred at room temperature overnight (~12 hours). After completion of the reaction, the mixture was poured into water (500 mL) and the aqueous phase was extracted three times with ethyl acetate (EA). The organic phases were combined, dried with anhydrous sodium sulfate (Na2SO4), filtered and concentrated under reduced pressure to give methyl 2-methyl-5-nitrobenzoate (15.3 g, 94% yield) as a yellow solid. The product was confirmed by 1H NMR (400 MHz, CDCl3): δ 8.52 (s, 1H), 8.05 (s, 1H), 7.30 (s, 1H), 3.83 (s, 3H), 2.56 (s, 3H). ESI-MS analysis showed m/z 196 [M + H]+.