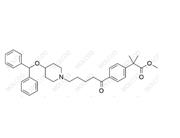
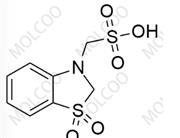
Aprocitentan Impurity 885702-34-1
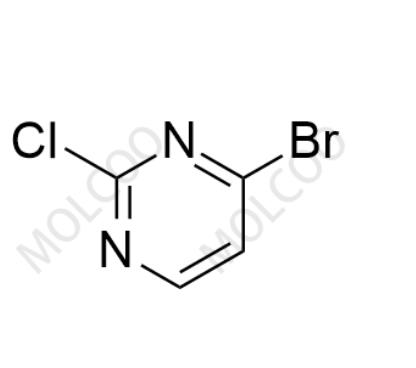
Product Number: A109010
English Name: Aprocitentan Impurity 10
English Alias: 4-bromo-2-chloropyrimidine
CAS Number: 885702-34-1
Molecular Formula: C₄H₂BrClN₂
Molecular Weight: 193.43
Product Advantages:
High-purity standard:With HPLC purity ≥99.0%, the structure is confirmed by 1H NMR, 13C NMR, and HRMS (high-resolution mass spectrometry), meeting the strict requirements of ICH Q2(R1) for impurity reference standards.
Reliable stability:Stable for 36 months when stored at -20°C in the dark, and with a degradation rate <0.5% after 10 days at room temperature in solution (e.g., dichloromethane), suitable for long-term storage and complex experimental conditions.
Precise traceability:As a characteristic impurity of halogenation reactions in aprocitentan synthesis, it accurately tracks the residue of brominating/chlorinating reagents and the degree of side reactions, assisting in locating process risk points.
Applications:
Drug quality research:Used for GC-MS or HPLC-MS detection of this impurity in aprocitentan APIs and formulations, controlling its content ≤0.1% in accordance with ICH Q3B standards to ensure drug safety.
Process development verification:In pyrimidine ring halogenation reactions, monitoring impurity content (e.g., reducing impurity from 0.8% to 0.1% when the bromine-chlorine reagent ratio is adjusted from 1:1.2 to 1:1.05) optimizes reaction parameters to reduce by-product formation.
Analytical method development:Serves as a reference material for establishing specific detection methods, such as system suitability verification of the GC-FID method (column DB-1701, temperature programming), ensuring method sensitivity and accuracy.
Toxicological research support:Provides reliable samples for evaluating potential impurity toxicity, facilitating the completion of genotoxic impurity (GTIs) risk assessment reports to meet regulatory requirements of FDA, EMA, etc.
Background Description:
Aprocitentan Impurity 10 is a dihalopyrimidine impurity introduced during aprocitentan (an angiotensin II receptor antagonist) synthesis due to excessive halogenating reagents or uncontrolled reaction temperature. The bromine and chlorine atoms in this impurity may participate in electrophilic substitution reactions, increasing the risk of metabolic toxicity in vivo. According to the ICH M7(R1) guideline, impurities with halogenated aromatic structures require strict genotoxicity screening, making their content control a key aspect of drug research and development. Currently, no specific limits for this impurity are defined in major pharmacopoeias, but industry standards generally reference ICH Q3A to set ≤0.1%.
Research Status:
Innovations in detection technology:The mainstream method is GC-MS using an HP-5MS capillary column (30m×0.25mm×0.25μm), helium as the carrier gas, and selected ion monitoring (SIM) mode to achieve a detection limit (LOD) of 0.01ppm. The HPLC-MS/MS method uses a C18 column (2.1×100mm, 3.5μm) with gradient elution, achieving a limit of quantitation (LOQ) of 0.05μg/mL for trace analysis.
Synthesis mechanism research:Impurity formation is closely related to the feeding order of brominating agents (e.g., N-bromosuccinimide) and chlorinating agents (e.g., phosphorus oxychloride). Studies show that adding chlorinating agents first followed by dropwise addition of brominating agents at ≤0°C reduces impurity formation by over 60%, while high temperatures (>60°C) significantly promote dihalogenation side reactions.
Safety assessment progress:In vitro Ames tests show no mutagenicity at concentrations ≤100μg/dish, but mild tubular epithelial cell clouding swelling was observed in high-dose groups (50mg/kg) during a 28-day repeated dosing test in rats, suggesting that reasonable limits (e.g., ≤0.08%) should be set based on toxicological data.






 China
China