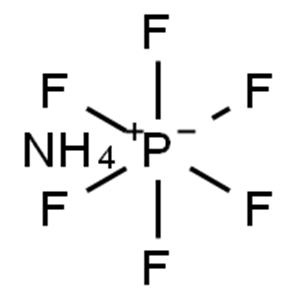
Ammonium hexafluorophosphate (chemical formula NH4PF6, CAS number 16941-17-2) is an inorganic ammonium salt,
belonging to the hexafluorophosphate class of fluorine-containing inorganic compounds.
It is an important raw material in lithium batteries, organic synthesis, and materials preparation.
Basic Properties:
Appearance:
White crystals or powder, with some hygroscopicity;
Solubility:
Easily soluble in water, soluble in methanol, ethanol, and some other organic solvents;
Hazards:
Class 8 corrosive hazardous chemicals, posing a serious risk of burns to skin and eyes.
Harmful if inhaled, ingested, or comes into contact with skin;
decomposes readily in the presence of strong alkalis, releasing ammonia gas;
contact with alkaline substances should be avoided.
Main Uses
Lithium-ion Battery Electrolyte:
One of its core uses is as an electrolyte raw material/additive for the preparation of high-purity lithium hexafluorophosphate (the mainstream electrolyte for lithium batteries), improving battery conductivity and cycle stability.
Organic Synthesis:
Used as a fluorinating agent and fluorine substitute, participating in the synthesis reactions of fluorine-containing organic compounds.
Materials Preparation:
Used to synthesize other hexafluorophosphates (such as potassium hexafluorophosphate and sodium hexafluorophosphate), and can also be applied in ionic liquids, catalysts, semiconductors, and photovoltaic materials.
Safety Precautions
Wear corrosion-resistant gloves, goggles, and protective clothing during operation.
Avoid direct contact with skin and eyes and inhalation of dust.
Store in a sealed, moisture-proof container, away from strong alkalis, oxidizers, and sources of ignition.
Transport, store, and manage according to relevant regulations for hazardous chemicals.




 China
China
