Product Number: A047058
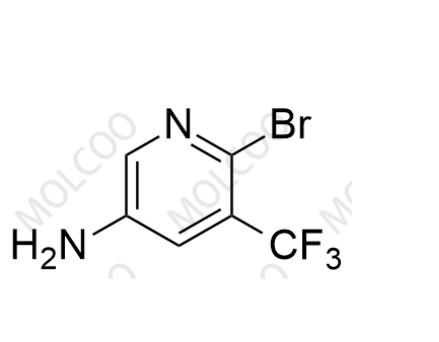
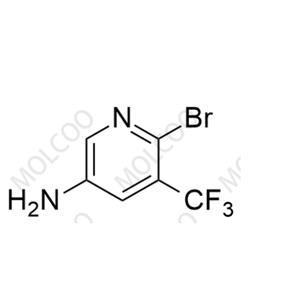
English Name: Apalutamide Impurity 58
English Alias: 6-bromo-5-(trifluoromethyl)pyridin-3-amine
CAS Number: 1642844-33-4
Molecular Formula: C6H4BrF3N2
Molecular Weight: 241.01
Advantages: Apalutamide Impurity 58 is synthesized via optimized processes and multi-stage purification, featuring ultra-high purity and consistent batch quality. Verified by HRMS and NMR, its precise chemical structure and residual impurity levels meet strict industry standards, providing a reliable reference for drug R&D and quality control to ensure accurate and reproducible experimental data.
Applications: Primarily used for impurity analysis, quality control, and safety assessment of Apalutamide. In R&D, it determines impurity content and evaluates impacts on efficacy and toxicity; in production, it serves as a key reference to monitor impurity levels, ensuring compliance with international regulations like ICH. It also applies to drug stability studies and degradation pathway analysis.
Background Description: As a critical prostate cancer treatment, impurity control for Apalutamide is central to its quality system. With stricter regulatory limits on drug impurities, research on Apalutamide Impurity 58 becomes essential for ensuring drug safety. Its presence may affect drug purity and efficacy, making precise characterization and control clinically significant.
Research Status: Current research focuses on developing highly sensitive detection methods and exploring generation mechanisms. Researchers use UPLC-MS/MS for trace analysis and optimize synthesis routes to reduce its formation during production. Meanwhile, studies on its effects on drug stability and biological activity are advancing, providing theoretical support for impurity control strategies.
WhatsAPP: +86 17320513646
E-mail: anna@molcoo.com
NOTE!
We can also customize related analogues and modified peptides including HPLC, MS, 1H-NMR, MS, HPLC, IR, UV, COA, MSDS.
This product is intended for laboratory use only!
WhatsAPP: +86 17320513646
E-mail: anna@molcoo.com
NEW IN STOCK!
The Molcoo Laboratory added drug impurity reference standards, including Baricitinib, Piperazine, Benzylpenicillin, Tranilast and multiple N-Nitroso drug impurities! Now available for immediate delivery!





 China
China