Product Number: A047052
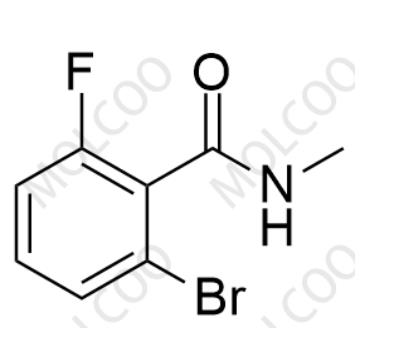
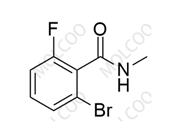
English Name: Apalutamide Impurity 52
English Alias: 2-bromo-6-fluoro-N-methylbenzamide
CAS Number: 1436306-43-2
Molecular Formula: C8H7BrFNO
Molecular Weight: 232.05
Advantages: Apalutamide Impurity 52 is manufactured using advanced synthesis techniques and stringent purification processes. This ensures an extremely high level of purity and excellent stability, with consistent quality across batches. The product undergoes rigorous structural confirmation and content determination through sophisticated analytical methods such as high-resolution mass spectrometry (HRMS) and nuclear magnetic resonance (NMR). With minimal impurity residues, it serves as a highly reliable reference standard for various applications in drug research, development, and quality control, significantly enhancing the accuracy and reliability of experimental data.
Applications: Primarily applied in the impurity research, quality analysis, and control of Apalutamide drugs. During the drug development process, it is used to accurately determine the content of this impurity in Apalutamide, helping to assess its potential impact on the drug's safety and efficacy. In pharmaceutical production, as a key reference substance, it enables precise detection of the impurity, ensuring that the final product meets or exceeds international quality standards and regulatory requirements, such as those set by the ICH. Additionally, it plays a vital role in drug stability studies and the investigation of degradation pathways.
Background Description: Apalutamide is a crucial drug in the treatment of prostate cancer, and maintaining strict control over its impurities is essential for ensuring drug quality and patient safety. As regulatory authorities worldwide impose increasingly stringent requirements on drug impurity limits, in-depth research on impurities like Apalutamide Impurity 52 has become an integral part of the pharmaceutical development process. The presence of such impurities can potentially affect the drug's purity, stability, and therapeutic effects, making their identification, characterization, and control critical for the successful development and production of high-quality Apalutamide drugs.
Research Status: Currently, research efforts regarding Apalutamide Impurity 52 are focused on developing more sensitive and efficient detection methods. Scientists are leveraging advanced analytical techniques, such as ultra-high-performance liquid chromatography-tandem mass spectrometry (UPLC-MS/MS) and high-performance capillary electrophoresis (HPCE), to achieve trace-level detection and accurate quantification of the impurity. Simultaneously, studies are underway to elucidate its formation mechanisms during the drug synthesis process, as well as its interactions with the active pharmaceutical ingredients of Apalutamide. These investigations aim to provide comprehensive theoretical and technical support for optimizing drug production processes, enhancing impurity control strategies, and ultimately improving the overall quality of Apalutamide drugs.
WhatsAPP: +86 17320513646
E-mail: anna@molcoo.com
NOTE!
We can also customize related analogues and modified peptides including HPLC, MS, 1H-NMR, MS, HPLC, IR, UV, COA, MSDS.
This product is intended for laboratory use only!
WhatsAPP: +86 17320513646
E-mail: anna@molcoo.com
NEW IN STOCK!
The Molcoo Laboratory added drug impurity reference standards, including Baricitinib, Piperazine, Benzylpenicillin, Tranilast and multiple N-Nitroso drug impurities! Now available for immediate delivery!





 China
China