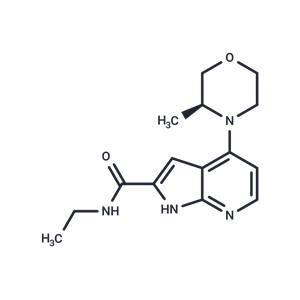
| Name | BAY-707 |
| Description | BAY-707, a highly potent and selective substrate-competitive inhibitor of MTH1 (NUDT1) with an IC50 of 2.3 nM, is well-tolerated in mice and exhibits a favorable pharmacokinetic (PK) profile compared to other MTH1 compounds. However, it demonstrates a clear lack of anticancer efficacy both in vitro and in vivo[1]. |
| In vitro | BAY-707 exhibits superior cellular target engagement with an EC50 of 7.6 nM, reflecting higher enzymatic potency (IC50=2.3 nM)[1] and demonstrates high cell permeability in the Caco-2 assay with an efflux ratio of 288 nm/s[1]. BAY-707 (0-30 μM; 24 hours) shows no antiproliferative effects in HMEC, HeLa, and SW-480 cells[1]. It has a favorable physicochemical profile and promising in vitro pharmacokinetic properties, with high metabolic stability in both human microsomes (0.29 L/h/kg, Fmax=78%) and rat hepatocytes (0.54 L/h/kg, Fmax=87%)[1]. |
| In vivo | BAY-707 (orally adminstation; 50-250 mg/kg; 2 weeks) is well-tolerated in nude mice, after 7-days treatment, body weight loss does not exceed 10% [1] and it also exhibits superior biochemical potency, cellular target engagement, and a pharmacokinetic profile to other MTH1 tool compounds. However, Bay-077 exerts no anticancer efficacy either in mono- or in combination therapies in CT26 and NCI-H460 mice model[1]. |
| Storage | Shipping with blue ice/Shipping at ambient temperature. |
| Keywords | MTH1/NUDT1 | BAY-707 | BAY707 | BAY 707 |
| Inhibitors Related | 5-Fluorouracil | Adenine hemisulfate | Erythromycin thiocyanate | Guanidine hydrochloride | Hexane-1,6-diol | 1,4-Naphthoquinone | Adenine | Vidarabine | Carbazole | Thymidine | Docosanoic acid | Usnic Acid |

 United States
United States