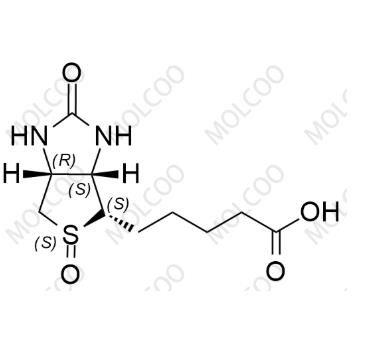
Biotin (S)-Sulfoxide
Product Code:B042023
English Name:Biotin (S)-Sulfoxide
English Alias:5-((3aS,4S,5S,6aR)-5-oxido-2-oxohexahydro-1H-thieno[3,4-d]imidazol-4-yl)pentanoic acid
CAS No.:10406-89-0
Molecular Formula:C₁₀H₁₆N₂O₄S
Molecular Weight:260.31
High-Purity Guarantee:Confirmed by HPLC (≥99.0%), combined with multiple techniques such as NMR (1H, 13C), HRMS, and elemental analysis, providing reliable reference materials for biotin-related research.
Good Stability:Stable for 36 months at -20℃ under light-protected, sealed storage; degradation rate <0.3% in common solvent systems like methanol - water within 6 months, ensuring the stability and reproducibility of experimental data.
Quality Control Testing:Used for UPLC-MS/MS detection of Biotin (S)-Sulfoxide impurity in biotin API and formulations, strictly controlling impurity content to meet ICH Q3A standards (single impurity limit ≤0.1%) and ensuring drug quality.
Drug Research and Development:During the development of biotin-based drugs, it serves as a reference substance to study its impact on drug activity and stability, helping to optimize the synthesis process and improve drug quality.
Metabolism Research:As a possible oxidation product of biotin in vivo, it is used to study the metabolic pathways and mechanisms of biotin, facilitating the understanding of drug transformation processes in the body.
Biotin, a water-soluble vitamin, plays a crucial role in various physiological processes. It is commonly used to treat conditions such as hair loss and brittle nails and is widely applied in biomedical research and pharmaceutical formulations. Biotin (S)-Sulfoxide, as an oxidation impurity of biotin, may be generated during biotin production and storage due to exposure to oxygen, light, or high temperatures. The sulfoxide group in its structure can change the molecular polarity and chemical properties, potentially affecting the quality, stability, and safety of biotin products. With the increasing requirements for drug impurity control in the pharmaceutical industry, the study of this impurity has become increasingly important.
Detection Technology:UPLC-MS/MS with a C18 column (1.7μm) and 0.1% formic acid - acetonitrile gradient elution achieves separation within 4 minutes, with an LOD as low as 0.003 ng/mL, enabling precise detection of trace impurities.
Formation Mechanism:Studies have shown that in an aerobic environment, especially under conditions of high temperature, light, or metal ion catalysis, the sulfur atom of biotin is easily oxidized to form Biotin (S)-Sulfoxide. By optimizing the production process, such as using inert gas protection and storing in the dark at low temperatures, the formation of this impurity can be effectively inhibited.
Safety Evaluation:In vitro cytotoxicity tests show that the IC₅₀ of this impurity against HepG2 cells is 198.5 μM (biotin has no obvious toxic concentration at tested levels). Although the toxicity is relatively low, its content in drugs still needs to be strictly controlled. Currently, long-term stability tests are being carried out to systematically study its degradation patterns under different humidity, light, and temperature conditions.
WhatsAPP: +86 17320513646
E-mail: anna@molcoo.com
NOTE!
We can also customize related analogues and modified peptides including HPLC, MS, 1H-NMR, MS, HPLC, IR, UV, COA, MSDS.
This product is intended for laboratory use only!
WhatsAPP: +86 17320513646
E-mail: anna@molcoo.com
NEW IN STOCK!
The Molcoo Laboratory added drug impurity reference standards, including Baricitinib, Piperazine, Benzylpenicillin, Tranilast and multiple N-Nitroso drug impurities! Now available for immediate delivery!