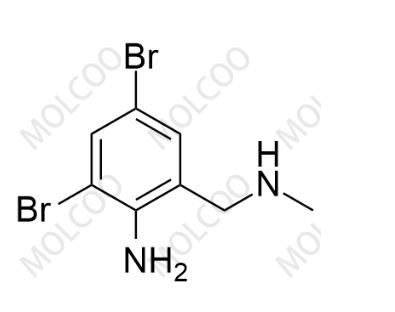
Product Number: B008036
English Name: Bromhexine Impurity 36
English Alias: 2,4-dibromo-6-((methylamino)methyl)aniline
CAS Number: 740758-41-2
Molecular Formula: C₈H₁₀Br₂N₂
Molecular Weight: 293.99
As an impurity reference standard for bromhexine, this compound has the following advantages:
Well-defined structure and high stability, which can be used to analyze the by-product formation mechanism of bromination and amination reactions in bromhexine synthesis, optimizing processes to control dibromo impurity generation;
As a reference standard containing bromine atoms and amino groups, it provides a standard substance for detecting halogenated aromatic amine impurities in drugs, improving the quantitative accuracy of methods such as HPLC;
Helps study the impact of bromine substituents on drug stability and toxicological properties (halogenated aromatic amines may have potential genotoxicity) to provide a scientific basis for impurity control strategies.
Drug Development: Used as an impurity reference standard to identify and quantify Impurity 36 in bromhexine preparations, evaluating the purity of APIs and formulations;
Quality Control: Acting as a standard substance to validate the sensitivity of detection methods (e.g., HPLC or LC-MS), ensuring the impurity content meets pharmacopoeia requirements during production;
Toxicological Research: Assisting in evaluating the potential genotoxicity of halogenated aromatic amine impurities to provide data support for drug safety evaluation.
Bromhexine is a mucolytic drug used for treating respiratory diseases. If the brominating reagent is excessive or the amination reaction conditions are out of control during its synthesis, dibromo impurities such as 2,4-dibromo-6-((methylamino)methyl)aniline are easily generated. Since halogenated aromatic amine compounds may have potential carcinogenic risks, research on this impurity is an important part of bromhexine quality control and safety assessment.
Current research focuses on:
Synthesis Methods: Developing high-purity synthesis processes for Impurity 36 to solve the purification challenges of halogenated aromatic amines and meet the needs of toxicological research;
Detection Technologies: Establishing trace detection methods (detection limits reach ppb level) for this impurity using ultra-high-performance liquid chromatography-tandem mass spectrometry (UPLC-MS/MS) technology;
Toxicological Evaluation: Studying the potential mutagenicity of bromine substituents through in vitro Ames tests and animal models;
Process Control: Analyzing the inducements (such as bromine dosage, reaction temperature)





 China
China