Cilnidipine Impurity 1;146845-34-3
=
WhatsAPP: +86 17386083646
E-mail: anna@molcoo.com
Product Number: C050001
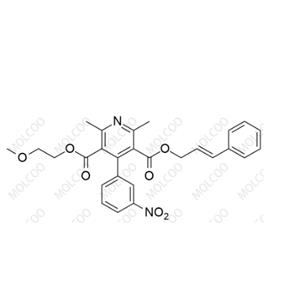
English Name: Cilnidipine Impurity 1
English Alias: 3-cinnamyl 5-(2-methoxyethyl) 2,6-dimethyl-4-(3-nitrophenyl)pyridine-3,5-dicarboxylate
CAS Number: 146845-34-3
Molecular Formula: C₂₇H₂₆N₂O₇
Molecular Weight: 490.5
As an important impurity of cilnidipine, the study of Cilnidipine Impurity 1 has significant advantages: It helps to analyze the synthesis process of cilnidipine, identify the source of impurities, optimize the production process, and improve the purity of the main drug. It can be used as a reference standard for quality inspection to accurately control the content of this impurity in drugs and ensure the stability of drug quality. In-depth research on it can also clarify the potential impact of impurities on the safety and effectiveness of drugs, providing a scientific basis for drug research, development, and supervision.
It is mainly applied in the research, development, and quality control of cilnidipine-related drugs. In the research and development stage, as an impurity reference standard, it assists researchers in identifying and quantifying this impurity in drugs, evaluating drug quality and safety. In the quality control process, as a standard substance, it is used to verify the accuracy and reliability of impurity detection methods, ensuring that the quality of drugs meets the standards in all links from production to circulation, and facilitating the formulation and improvement of drug quality standards.
Cilnidipine, a calcium channel blocker, is commonly used in the treatment of cardiovascular diseases such as hypertension and is widely applied in clinical treatment. With the increasing requirements for drug quality and safety, impurity research has become a crucial part of drug research, development, and production processes. Cilnidipine Impurity 1, as an impurity that may be generated during the synthesis or storage of cilnidipine, its content and properties directly affect the quality of cilnidipine drugs. Therefore, the research on this impurity has received high attention from the pharmaceutical industry.
Currently, research on Cilnidipine Impurity 1 covers multiple aspects. In the field of synthesis, researchers are committed to exploring more efficient, low-cost, and high-purity synthesis methods to obtain sufficient amounts of impurities for subsequent research. In terms of analytical detection technologies, advanced techniques such as high-performance liquid chromatography (HPLC) and liquid chromatography-mass spectrometry (LC-MS) are continuously being adopted to improve the detection sensitivity and accuracy of this impurity. In toxicological research, animal experiments and in vitro cell experiments are used to evaluate the potential toxicity of this impurity to humans, providing data support for determining the safe limit of impurities. At the same time, research on the formation mechanism and control strategies of this impurity during the production process of cilnidipine is also being continuously and deeply promoted.
NOTE!
We can also customize related analogues and modified peptides including HPLC, MS, 1H-NMR, MS, HPLC, IR, UV, COA, MSDS.
This product is intended for laboratory use only!
WhatsAPP: +86 17386083646
E-mail: anna@molcoo.com
NEW IN STOCK!
The Molcoo Laboratory added drug impurity reference standards, including Baricitinib, Piperazine, Benzylpenicillin, Tranilast and multiple N-Nitroso drug impurities! Now available for immediate delivery!





 China
China


