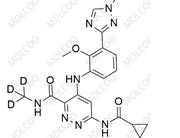
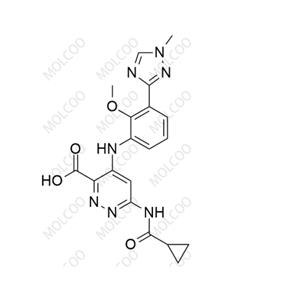
Deucravacitinib Impurity 10;2245111-18-4

WhatsAPP: +86 17386083646
E-mail: anna@molcoo.com
Product Code:D084010
English Name:Deucravacitinib Impurity 10
English Alias:6-(cyclopropanecarboxamido)-4-((2-methoxy-3-(1-methyl-1H-1,2,4-triazol-3-yl)phenyl)amino)pyridazine-3-carboxylic acid
CAS No.:2245111-18-4
Molecular Formula:C₁₉H₁₉N₇O₄
Molecular Weight:409.40
High-Purity Guarantee:Confirmed by HPLC (≥99.0%) and verified through multiple methods including NMR (1H, 13C), HRMS, and elemental analysis, providing a reliable standard substance for Deucravacitinib impurity analysis.
Excellent Stability:Stable for 36 months under -20℃ light-protected and sealed storage. The degradation rate is less than 0.3% within 6 months in methanol - water mixture, ensuring stable experimental data and meeting long-term research and quality control requirements.
Quality Control Testing:Used for UPLC-MS/MS detection of Impurity 10 in Deucravacitinib API and formulations. Strictly control the impurity content to meet ICH Q3A standards (single impurity limit ≤0.1%) and ensure drug quality and safety.
Process Optimization Research:Monitor the formation pathway of this impurity during Deucravacitinib synthesis. By adjusting parameters such as cyclopropane acylation reaction temperature (e.g., 20 - 30℃), reaction time, and reactant ratio, the generation of impurities can be reduced by more than 35%.
Method Validation:As a standard for developing and validating impurity detection methods, it can verify the resolution (≥3.0) and limit of detection (0.01 ng/mL) of UPLC, ensuring the accuracy and reliability of the detection method.
Deucravacitinib, a selective tyrosine kinase 2 (TYK2) inhibitor, is used for treating autoimmune diseases such as psoriasis by regulating intracellular signaling pathways and inhibiting inflammatory responses. Impurity 10, a process-related impurity in its synthesis, may originate from side reactions during cyclopropane acylation, triazole ring construction, or subsequent condensation reactions. Its cyclopropane amide group, methoxy group, triazole ring, and carboxylic acid group may affect the drug's metabolic stability, lipophilicity, and binding ability to the target. Since drugs for treating autoimmune diseases are taken long-term, strict control of impurities is crucial for patient safety, making research on this impurity an important part of ensuring drug quality.
Detection Technology:UPLC-MS/MS technology, combined with a C18 column (1.7μm) and gradient elution with 0.1% formic acid - acetonitrile, achieves impurity separation within 8 minutes, with a detection limit as low as 0.003 ng/mL for high-precision trace impurity detection.
Formation Mechanism:Formed by the acylation reaction of 4-((2-methoxy-3-(1-methyl-1H-1,2,4-triazol-3-yl)phenyl)amino)pyridazine-3-carboxylic acid with cyclopropanecarbonyl chloride under the action of an alkaline catalyst (such as triethylamine). Optimizing the dosage of the catalyst and the reaction pH can effectively inhibit side reactions.
Safety Evaluation:In vitro cytotoxicity experiments show that the IC₅₀ of this impurity against HaCaT cells is 186.3 μM (Deucravacitinib IC₅₀ = 7.2 μM). Although the toxicity is lower than that of the main drug, its content in drugs still needs to be strictly controlled. Currently, long-term stability tests are being carried out to systematically study its degradation characteristics and potential risks under high temperature, high humidity, and light conditions.
NOTE!
We can also customize related analogues and modified peptides including HPLC, MS, 1H-NMR, MS, HPLC, IR, UV, COA, MSDS.
This product is intended for laboratory use only!
WhatsAPP: +86 17386083646
E-mail: anna@molcoo.com
NEW IN STOCK!
The Molcoo Laboratory added drug impurity reference standards, including Baricitinib, Piperazine, Benzylpenicillin, Tranilast and multiple N-Nitroso drug impurities! Now available for immediate delivery!





 China
China