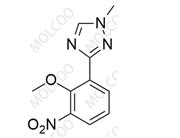
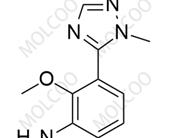
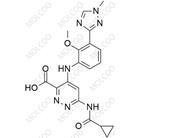
Deucravacitinib Impurity ;1609392-27-9
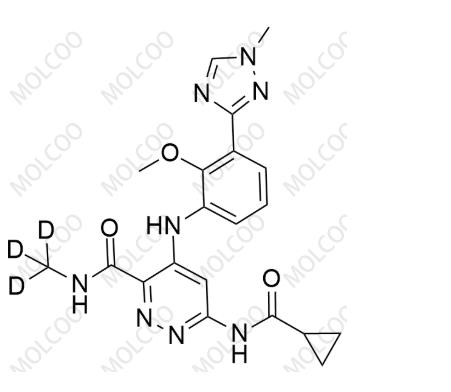
Product Code:D084000
English Name:Deucravacitinib
English Alias:6-(cyclopropanecarboxamido)-4-((2-methoxy-3-(1-methyl-1H-1,2,4-triazol-3-yl)phenyl)amino)-N-(methyl-d3)pyridazine-3-carboxamide
CAS No.:1609392-27-9
Molecular Formula:C₂₀H₁₉D₃N₈O₃
Molecular Weight:425.46
High-Purity Guarantee:Confirmed by HPLC (≥99.5%), combined with multiple techniques such as NMR (1H, 13C, including deuterium signal analysis), HRMS, and elemental analysis, providing accurate reference standards for drug research, development, and quality control.
Excellent Stability:Stable for 36 months at -20℃ under light-protected, sealed storage; degradation rate <0.2% in common solvents (such as DMSO, acetonitrile - water system) within 6 months, ensuring reliable experimental data.
Drug Development:As a novel tyrosine kinase 2 (TYK2) inhibitor, it is used in the development of drugs for autoimmune diseases such as psoriasis. It regulates the immune response and reduces the release of inflammatory factors by inhibiting the TYK2 signaling pathway.
Preclinical Research:In pharmacokinetic studies, its deuterated structure is utilized to explore the metabolic pathways and elimination patterns in vivo, optimizing dosing regimens; in pharmacodynamic studies, it assesses the therapeutic effects on disease models.
Quality Control:Used for UPLC-MS/MS detection of Deucravacitinib in active pharmaceutical ingredients and formulations, controlling impurity content to meet ICH Q3A and Q3B standards, ensuring drug quality and safety.
Deucravacitinib is an innovative oral small-molecule drug targeting tyrosine kinase 2 (TYK2). Autoimmune diseases such as psoriasis and rheumatoid arthritis are closely related to the abnormal activation of signaling pathways within immune cells. As a key signaling molecule, TYK2 is involved in the signal transduction of cytokines such as interferons and interleukins. Traditional therapeutic drugs have limitations in efficacy and significant side effects. Deucravacitinib specifically binds to the regulatory domain of TYK2, blocking its activation and avoiding widespread inhibition of other kinases, thus enabling more precise and safe immune regulation and bringing new directions for the treatment of autoimmune diseases.
Clinical Application Progress:Approved for the treatment of psoriasis. Data from multiple Phase III clinical trials show that it significantly improves patients' skin symptoms and quality of life compared with placebo; clinical trials for rheumatoid arthritis, systemic lupus erythematosus, and other diseases are ongoing to explore broader indications.
In-Depth Mechanism Research:The binding mode of Deucravacitinib to the TYK2 protein is analyzed by cryo-electron microscopy to clarify the molecular mechanism of its highly selective inhibition; gene editing techniques are used to knock down or overexpress TYK2 to further verify its regulatory effect on downstream signaling pathways.
Formulation Optimization:Research on new formulation technologies, such as nanoparticle delivery systems and sustained-release formulations, to improve drug bioavailability and patient compliance; evaluate the release characteristics and stability of different formulations in vitro and in vivo to provide better choices for clinical applications
WhatsAPP: +86 17320513646
E-mail: anna@molcoo.com
NOTE!
We can also customize related analogues and modified peptides including HPLC, MS, 1H-NMR, MS, HPLC, IR, UV, COA, MSDS.
This product is intended for laboratory use only!
WhatsAPP: +86 17320513646
E-mail: anna@molcoo.com
NEW IN STOCK!
The Molcoo Laboratory added drug impurity reference standards, including Baricitinib, Piperazine, Benzylpenicillin, Tranilast and multiple N-Nitroso drug impurities! Now available for immediate delivery!






 China
China