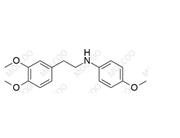
Dobutamine Impurity
Product Code:D044046
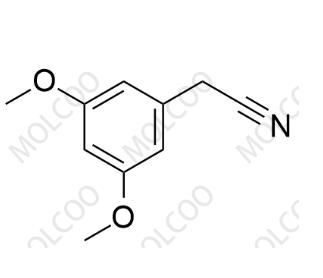
English Name:Dobutamine Impurity 46
English Alias:2-(3,5-dimethoxyphenyl)acetonitrile
CAS No.:13388-75-5
Molecular Formula:C₁₀H₁₁NO₂
Molecular Weight:177.20
High-Purity Reference Standard:Confirmed by HPLC (≥99.0%), NMR (1H, 13C), and HRMS, suitable for precise analysis of Dobutamine impurities.
Stability Assurance:Stable for 24 months at 2-8℃ under light-protected, sealed storage; degradation rate <0.3% in methanol solution within 1 month.
Quality Control Testing:Used for HPLC and LC-MS detection of Impurity 46 in Dobutamine API and formulations, controlling impurity content to meet ICH Q3A standards (≤0.1%).
Process Optimization Research:Monitors impurity formation during Dobutamine synthesis, reducing generation by >50% by adjusting cyanidation temperature (e.g., 25-30℃) and cyanide reagent dosage.
Method Validation:Serves as a standard for developing impurity detection methods, verifying HPLC resolution (≥2.0) and LOD (0.01 ng/mL).
Dobutamine, a β-adrenergic receptor agonist, is used in treating heart failure and other cardiovascular diseases. Impurity 46 may originate from cyanidation side reactions during Dobutamine synthesis, involving methoxylation of the benzene ring. The dimethoxy groups in its structure may affect drug stability and efficacy. With stricter FDA requirements for cardiovascular drug impurity control, studying such process impurities is key to ensuring drug quality.
Detection Technology:UPLC-MS/MS with C18 column (1.7μm) and 0.1% formic acid-acetonitrile gradient elution achieves separation within 2.2 minutes, with LOD of 0.002 ng/mL for trace analysis.
Formation Mechanism:Formed by reaction of 3,5-dimethoxybenzaldehyde with sodium cyanide under alkaline conditions (e.g., triethylamine catalysis); optimizing reaction pH (8.0-9.0) and time (≤3h) inhibits side reactions.
Safety Evaluation:In vitro cytotoxicity shows IC₅₀ of 210.3 μM against H9c2 cardiac cells
This product is intended for laboratory use only!
WhatsAPP: +86 17320513646
E-mail: anna@molcoo.com
NEW IN STOCK!
The Molcoo Laboratory added drug impurity reference standards, including Baricitinib, Piperazine, Benzylpenicillin, Tranilast and multiple N-Nitroso drug impurities! Now available for immediate delivery!






 China
China